Chapter 9: Q52E (page 286)
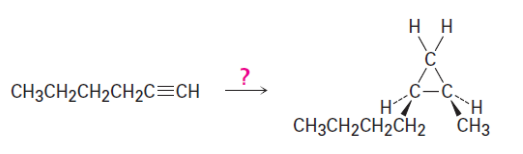
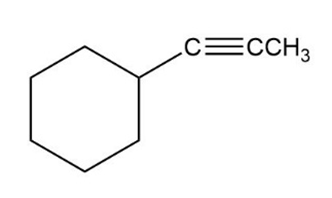
1-Octen-3-ol, a potent mosquito attractant commonly used in mosquito traps, can be prepared in two steps from hexanal, . The first step is an acetylide-addition reaction like that described in Problem 9-50. What is the structure of the product from the first step, and how can it be converted into 1-octen-3-ol?
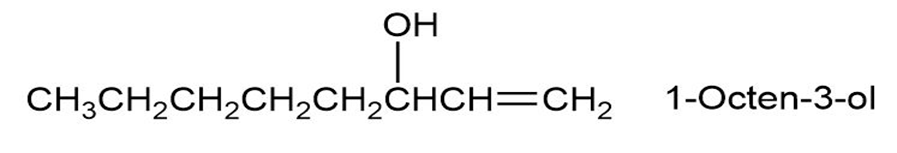
Short Answer
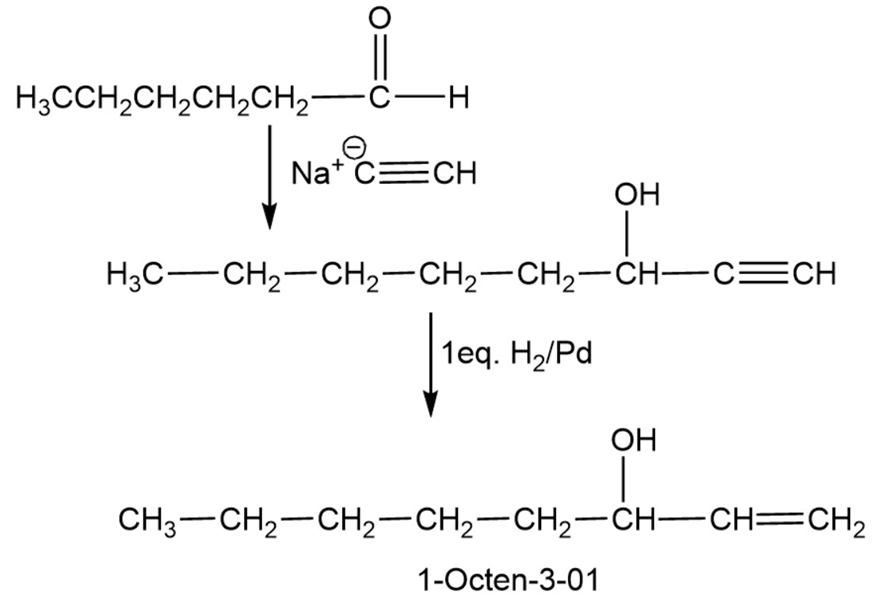
 Mechanism of reaction
Mechanism of reaction