Chapter 9: Q17E (page 263)
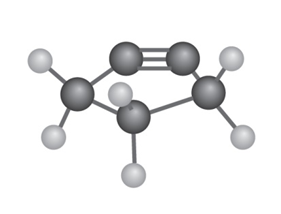
The following cycloalkyne is too unstable to exist. Explain
Short Answer
The linear geometry is distorted in the cyclopentyne and so the molecule is unstable.
Learning Materials
Features
Discover
Chapter 9: Q17E (page 263)
The following cycloalkyne is too unstable to exist. Explain

The linear geometry is distorted in the cyclopentyne and so the molecule is unstable.
All the tools & learning materials you need for study success - in one app.
Get started for free
Occasionally, a chemist might need to invert the stereochemistry of an alkene—that is, to convert a cis alkene to a trans alkene, or vice versa. There is no one-step method for doing an alkene inversion, but the transformation can be carried out by combining several reactions in the proper sequence. How would you carry out the following reactions?
(a)
(b)
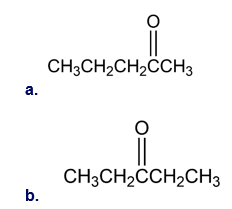
What alkynes would you start with to prepare the following ketones?
Let be a group and assume that for each positive integer i , Ni is a normal subgroup of G. If every element of can be written uniquely in the form ni1 .ni2 ...nikwith i1 < i2 < ... < ik and nijNij , prove that G Ni (see Exercise 34). [Hint: Adapt the proof of Theorem 9.1 by defining f(a1, a2...) to be the product of those ai that are not the identity element.]
There are seven isomeric alkynes with the formula . Draw and name them.
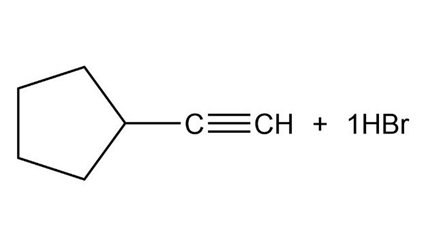
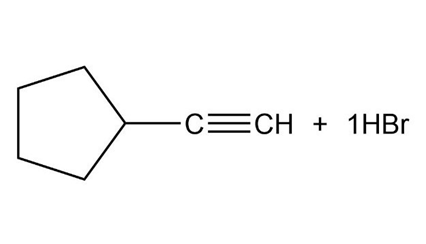
What products would you expect from the following reactions?
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.