Chapter 9: Q. 9-9-3P-b (page 265)
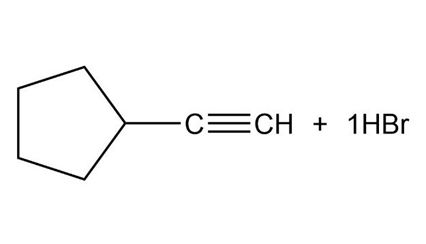
What products would you expect from the following reactions?
b.
Short Answer
Learning Materials
Features
Discover
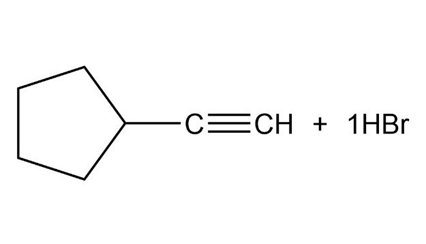
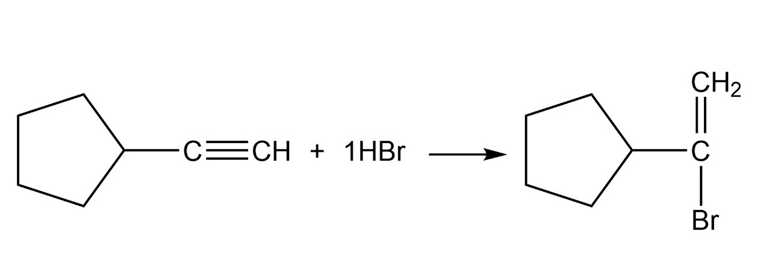
Chapter 9: Q. 9-9-3P-b (page 265)
What products would you expect from the following reactions?
b.



All the tools & learning materials you need for study success - in one app.
Get started for free
The sex attractant given off by the common housefly is an alkene named muscalure. Propose a synthesis of muscalure starting from acetylene and any alkyl halides needed. What is the IUPAC name for muscalure?
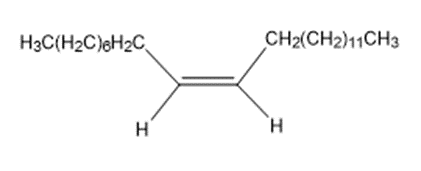
Muscalure
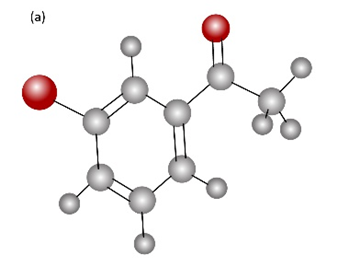
How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown Br)?
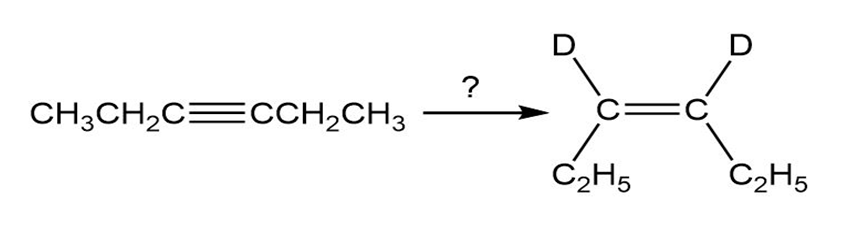
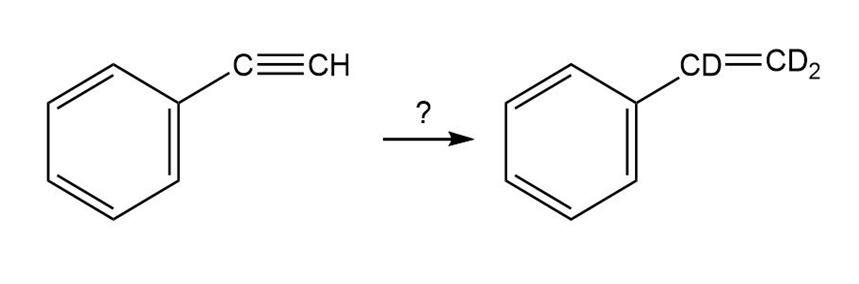
How would you carry out the following reactions to introduce deuterium into organic molecules?
(a)
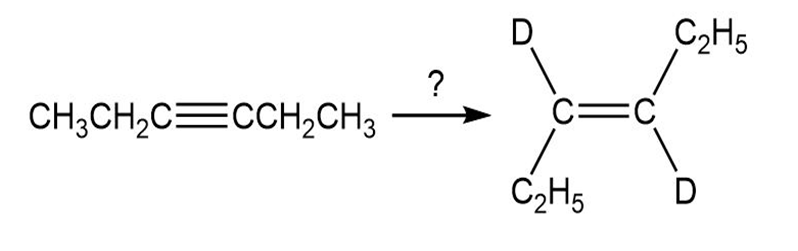
b.
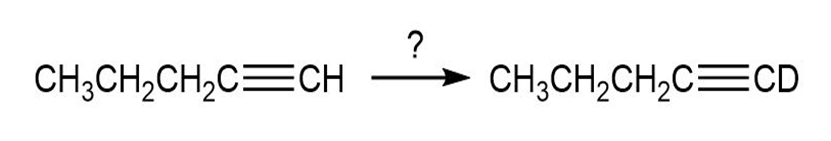
c.
d.
Synthesize the following compounds using 1-butyne as the only source of carbon along with any inorganic reagents you need. More than one step may be needed.(a) 1,1,2,2- Tetrachlorobutane(b) 1,1- Dichloro-2-ethylcyclopropane
A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. On hydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.
(a) How many degrees of unsaturation are present in the unknown structure?
(b) How many triple bonds are present?
(c) How many double bonds are present?
(d) How many rings are present?
(e) Draw a structure that fits the data.
What do you think about this solution?
We value your feedback to improve our textbook solutions.