Chapter 9: Q 38 E (page 286)
Question: Each of the following syntheses requires more than one step. How
would you carry them out?
(a)

b)
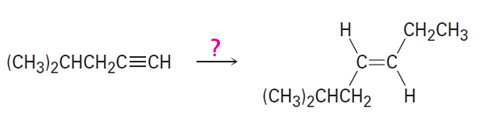
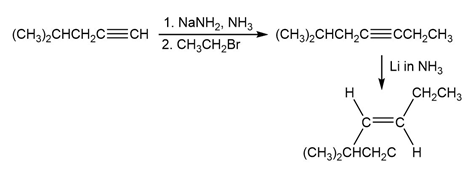
Short Answer
a)
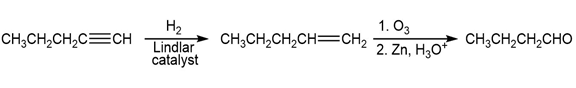
b)
Learning Materials
Features
Discover
Chapter 9: Q 38 E (page 286)
Question: Each of the following syntheses requires more than one step. How
would you carry them out?
(a)

b)

a)

b)

All the tools & learning materials you need for study success - in one app.
Get started for free
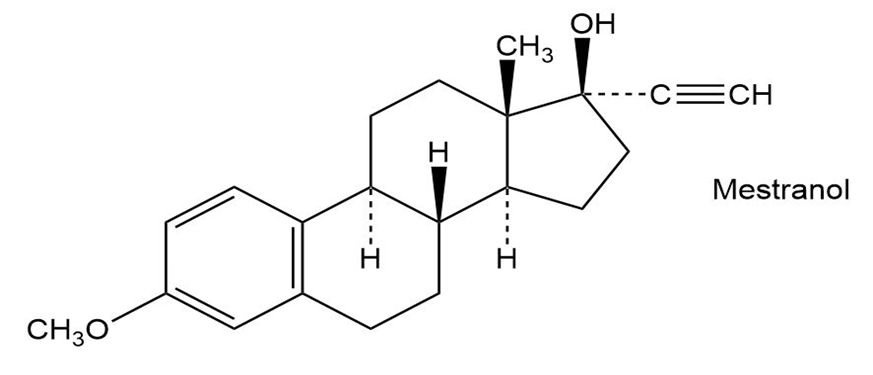
The oral contraceptive agent Mestranol is synthesized using a carbonyl addition reaction like that shown in Problem 9-50. Draw the structure of the ketone needed.
Using any alkyne needed, how would you prepare the following alkenes?
(b) cis-3-Heptene
The of acetone, of acetone, is 19.3. Which of the following bases is strong enough to deprotonate acetone?
(a)KOH (pkaof H2O 15.7)
A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. On hydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.
(a) How many degrees of unsaturation are present in the unknown structure?
(b) How many triple bonds are present?
(c) How many double bonds are present?
(d) How many rings are present?
(e) Draw a structure that fits the data.
The sex attractant given off by the common housefly is an alkene named muscalure. Propose a synthesis of muscalure starting from acetylene and any alkyl halides needed. What is the IUPAC name for muscalure?
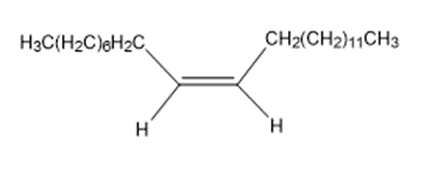
Muscalure
What do you think about this solution?
We value your feedback to improve our textbook solutions.