Chapter 17: Q45E (page 567)
How would you synthesize the following alcohols, starting with benzene and other alcohols of six or fewer carbons as your only organic reagents?
(a)
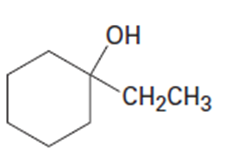
(b)
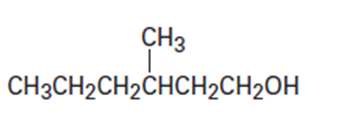
(c)
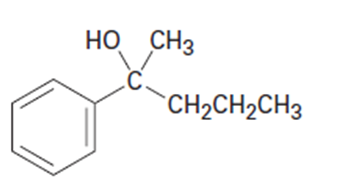
(d)
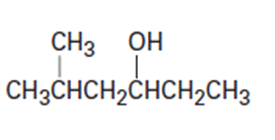
Short Answer
(a)
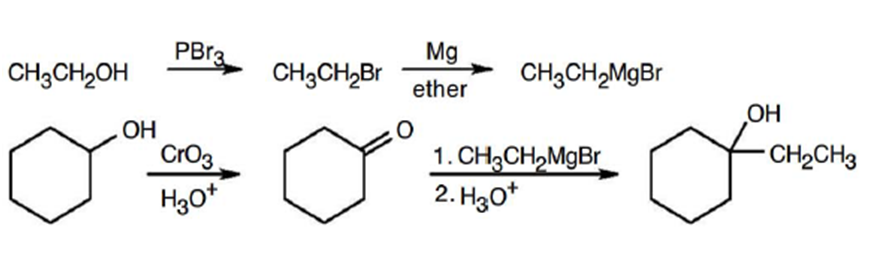
(b)
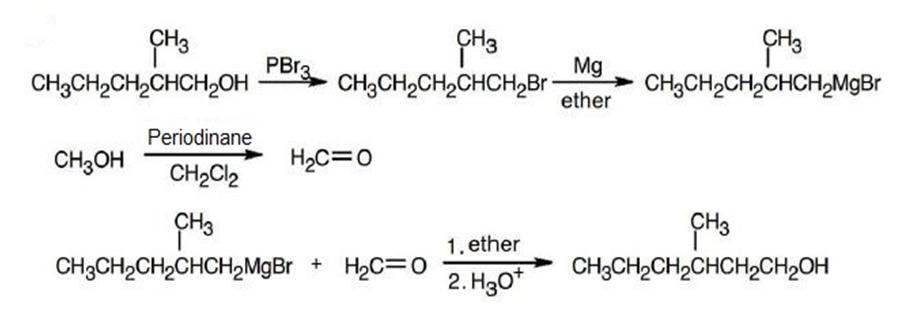
(c)
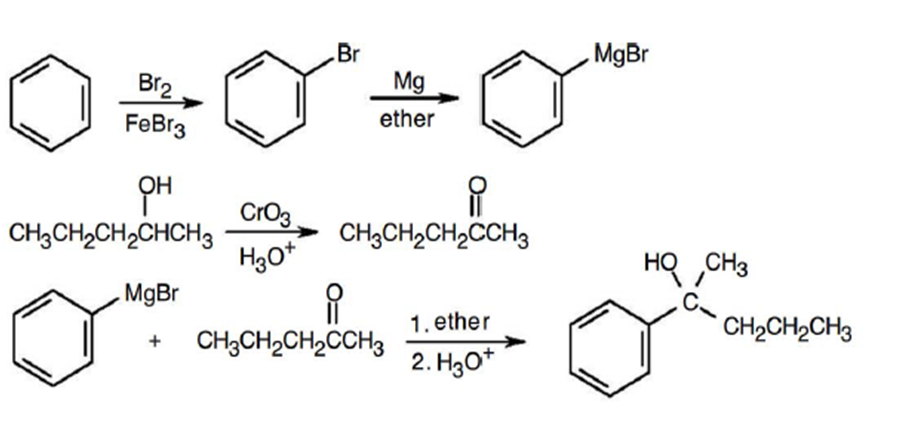
(d)
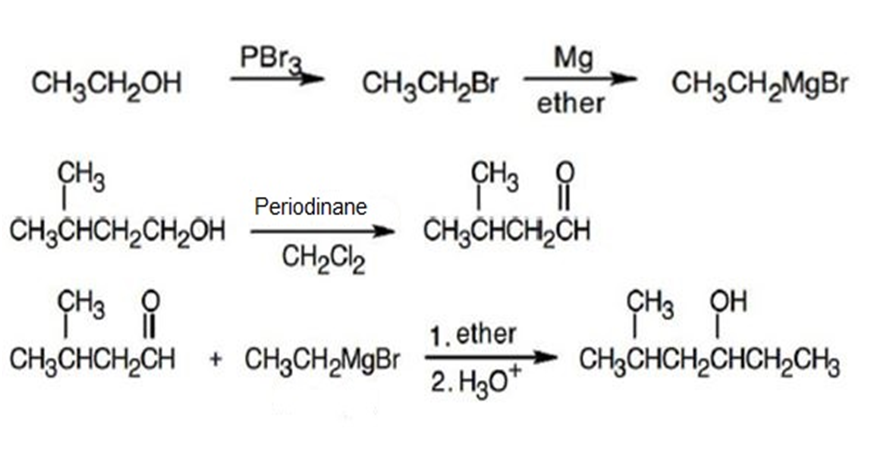
Learning Materials
Features
Discover
Chapter 17: Q45E (page 567)
How would you synthesize the following alcohols, starting with benzene and other alcohols of six or fewer carbons as your only organic reagents?
(a)

(b)

(c)

(d)

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
The following 1HNMR spectrum is that of an alcohol, C8H10O
. Propose a structure.
Question:Reaction of (S)-3-methyl-2-pentanone with methyl magnesium bromide followed by acidification yields 2,3-dimethyl-2-pentanol. What is the stereochemistry of the product? Is the product optically active?
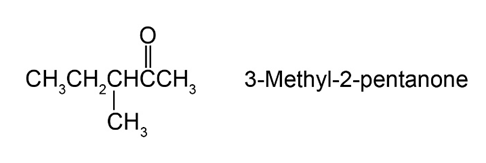
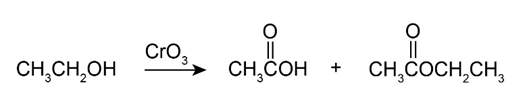
A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate:
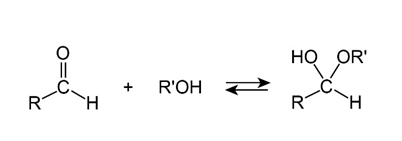
Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:
Name and assign R or S stereochemistry to the product (s) you would obtain by reaction of the following substance with ethylmagnesium bromide. Is the product chiral? Is it optically active? Explain.
Reduction of 2-butanone with NaBH4yields 2-butanol. Is the product chiral? Is it optically active? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.