Chapter 7: Q1P (page 291)
Name the following:

Short Answer
- IUPAC name: 5-bromo-pent-2-yne.
- IUPAC name: 3-bromo-2-chloro-oct-4-yne.
- IUPAC name: 1-methoxy-pent-2-yne.
- IUPAC name: 3-ethyl-hex-1-yne.
Learning Materials
Features
Discover
Chapter 7: Q1P (page 291)
Name the following:

All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following in order from strongest base to weakest base:
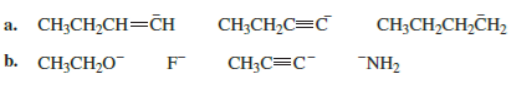
Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products?
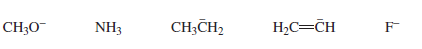
For each of the following alkynes, draw the products obtained from (1) the acid-catalyzed addition of water
(mercuric ion is added for part a) and from (2) hydroboration–oxidation:
a. 1-butyne b. 2-butyne c. 2-pentyne
Draw the mechanism for the following reaction
Do the equilibria of the following acid–base reactions lie to the right or the left? (The pKa of H2O2 is 11.6.)
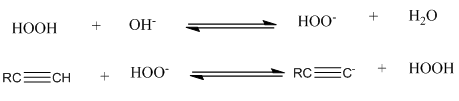
What do you think about this solution?
We value your feedback to improve our textbook solutions.