Chapter 7: Q24P (page 306)
Rank the following in order from strongest base to weakest base:
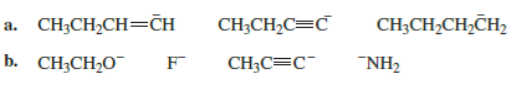
Short Answer
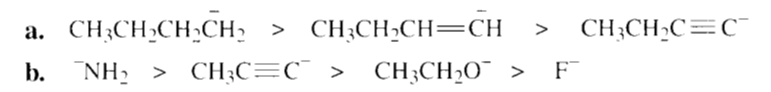
Learning Materials
Features
Discover
Chapter 7: Q24P (page 306)
Rank the following in order from strongest base to weakest base:
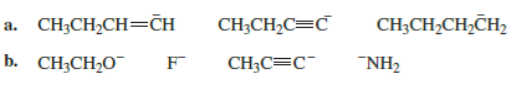
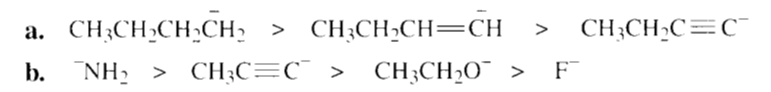
All the tools & learning materials you need for study success - in one app.
Get started for free
What is each compound’s systematic name?
Draw the mechanism for the following reaction
Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products?
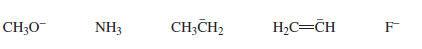
a. Starting with 3-methyl-1-butyne, how can you prepare the following alcohols?
1. 2-methyl-2-butanol
2. 3-methyl-1-butanol
b. In each case, a second alcohol would also be obtained. What alcohol would it be?
Drawing on what you know about the stereochemistry of alkene addition reactions:
a. Write the mechanism for the reaction of 2-butyne with one equivalent of Br2.
b. Predict the configuration of the product of the reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.