Chapter 7: Q43P (page 315)
a. Starting with 3-methyl-1-butyne, how can you prepare the following alcohols?
1. 2-methyl-2-butanol
2. 3-methyl-1-butanol
b. In each case, a second alcohol would also be obtained. What alcohol would it be?
Learning Materials
Features
Discover
Chapter 7: Q43P (page 315)
a. Starting with 3-methyl-1-butyne, how can you prepare the following alcohols?
1. 2-methyl-2-butanol
2. 3-methyl-1-butanol
b. In each case, a second alcohol would also be obtained. What alcohol would it be?
All the tools & learning materials you need for study success - in one app.
Get started for free
Drawing on what you know about the stereochemistry of alkene addition reactions:
a. Write the mechanism for the reaction of 2-butyne with one equivalent of Br2.
b. Predict the configuration of the product of the reaction.
How can the following compounds be synthesized, starting with a hydrocarbon that has the same number of carbons as the desired product?
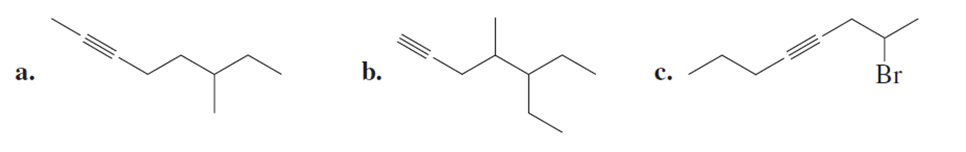
Name the following:
Name the following:
Why does cis-2-butene have a higher boiling point than trans-2-butene?
What do you think about this solution?
We value your feedback to improve our textbook solutions.