Chapter 7: Q47P (page 316)
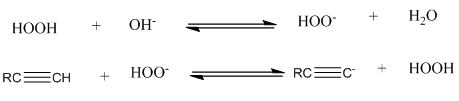
Do the equilibria of the following acid–base reactions lie to the right or the left? (The pKa of H2O2 is 11.6.)
Short Answer
The 1st equilibrium lies to the right but the 2nd equilibrium lies to the left.
Learning Materials
Features
Discover
Chapter 7: Q47P (page 316)
Do the equilibria of the following acid–base reactions lie to the right or the left? (The pKa of H2O2 is 11.6.)

The 1st equilibrium lies to the right but the 2nd equilibrium lies to the left.
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why sodium amide cannot be used to form a carbanion from an alkane in a reaction that favors products.
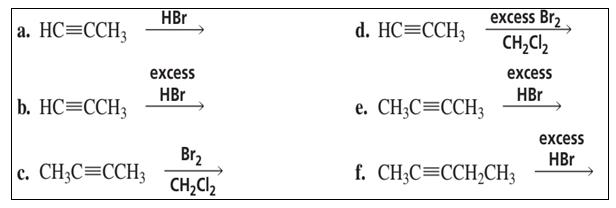
What is the major product of each of the following reactions?
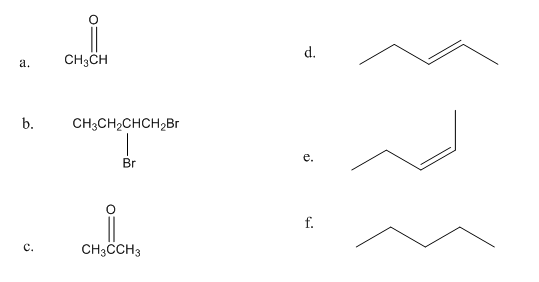
How can the following compounds be prepared using ethyne as the starting material?
Draw the structures and give the common and systematic names for the seven alkynes with molecular formula C6H10.
Describe the alkyne you would start with and the reagents you would use if you wanted to Synthesize
a. Pentane. b. Cis-2-butene. c. trans-2-pentene. d. 1-hexene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.