Chapter 7: Q19P (page 304)
Describe the alkyne you would start with and the reagents you would use if you wanted to Synthesize
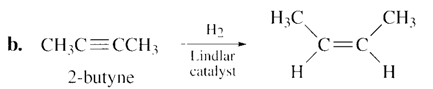
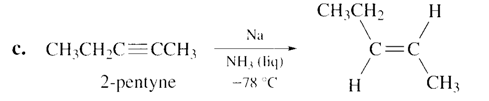
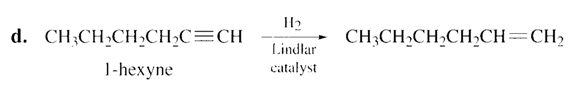
a. Pentane. b. Cis-2-butene. c. trans-2-pentene. d. 1-hexene.
Short Answer
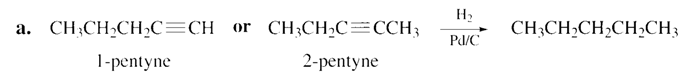
Learning Materials
Features
Discover
Chapter 7: Q19P (page 304)
Describe the alkyne you would start with and the reagents you would use if you wanted to Synthesize
a. Pentane. b. Cis-2-butene. c. trans-2-pentene. d. 1-hexene.
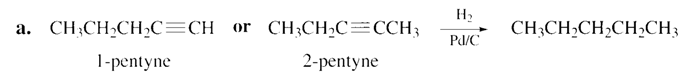



All the tools & learning materials you need for study success - in one app.
Get started for free
Which carbocation is more stable?
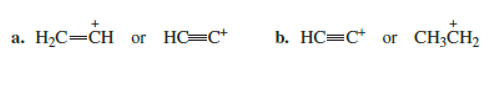
Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products?
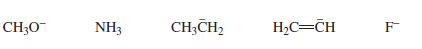
How can the following compounds be prepared using ethyne as the starting material?
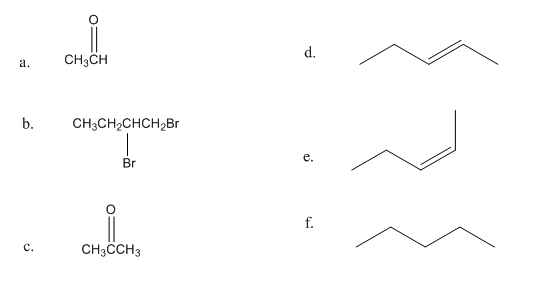
A student was given the structural formulas of several compounds and was asked to give them systematic names. How many did she name correctly? Correct those that are misnamed.
a. 4-ethyl-2-pentyne b. 1-bromo-4-heptyne c. 2-methyl-3-hexyne d. 3-pentyne
a. Starting with 3-methyl-1-butyne, how can you prepare the following alcohols?
1. 2-methyl-2-butanol
2. 3-methyl-1-butanol
b. In each case, a second alcohol would also be obtained. What alcohol would it be?
What do you think about this solution?
We value your feedback to improve our textbook solutions.