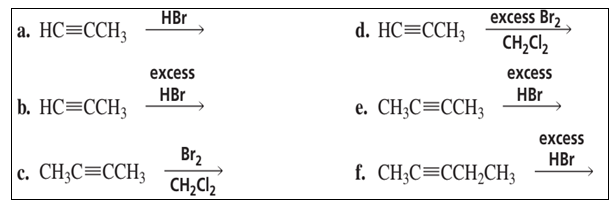
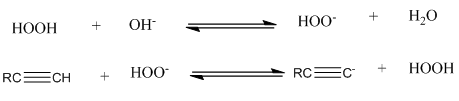
Chapter 7: Q31P (page 314)
A student was given the structural formulas of several compounds and was asked to give them systematic names. How many did she name correctly? Correct those that are misnamed.
a. 4-ethyl-2-pentyne b. 1-bromo-4-heptyne c. 2-methyl-3-hexyne d. 3-pentyne
Short Answer
Here we have to find out which names are correct:
Only C is correct.
a) 4-methyl-2-hexyne
b) 7-bromo-3-heptyne
c) Correct
d) 2-pentyne
This naming is given according to IUPAC nomenclature.