Chapter 7: Q45 P (page 315)
Which of the following pairs are keto–enol tautomers?
Short Answer
Only c and e are keto-enol tautomerism
Learning Materials
Features
Discover
Chapter 7: Q45 P (page 315)
Which of the following pairs are keto–enol tautomers?
Only c and e are keto-enol tautomerism
All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following bases can remove a proton from a terminal alkyne in a reaction that favors products?
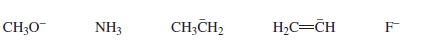
Draw the condensed and skeletal structures for each of the following:
a. 1-chloro-3-hexyne
b. cyclooctyne
c. isopropylacetylene
d. sec-butylisobutylacetylene
e. 4,4-dimethyl-1-pentyne
f. dimethylacetylene
Draw all the enol tautomers for each of the ketones in Problem 15.
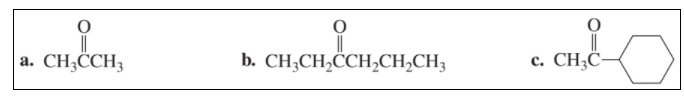
Name the following:
What ketones would be formed from the acid-catalyzed addition of water to 3-heptyne?
What do you think about this solution?
We value your feedback to improve our textbook solutions.