Chapter 7: Q14P (page 301)
What ketones would be formed from the acid-catalyzed addition of water to 3-heptyne?
Short Answer
R-C≡C-R' + H₂O → R-CH₂-CO-R' + R-CO-CH₂-R'
As here unsymmetrical alkyne is there so two ketones will form:
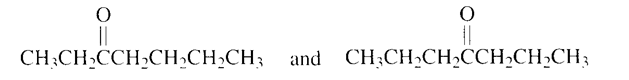
Learning Materials
Features
Discover
Chapter 7: Q14P (page 301)
What ketones would be formed from the acid-catalyzed addition of water to 3-heptyne?
R-C≡C-R' + H₂O → R-CH₂-CO-R' + R-CO-CH₂-R'
As here unsymmetrical alkyne is there so two ketones will form:
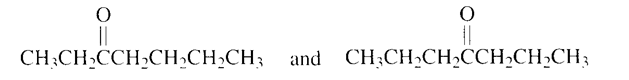
All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following names are correct? Correct those that are not correct.
a. 4-heptyne
b. 2-ethyl-3-hexyne
c. 4-chloro-2-pentyne
d. 2,3-dimethyl-5-octyne
e. 4,4-dimethyl-2-pentyne
f. 2,5-dimethyl-3-hexyne
Identify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.
Explain why sodium amide cannot be used to form a carbanion from an alkane in a reaction that favors products.
What is the molecular formula of a hydrocarbon that has 1 triple bond, 2 double bonds, 1 ring, and 32 carbons?
What is the molecular formula for a monocyclic hydrocarbon with 14 carbons and 2 triple bonds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.