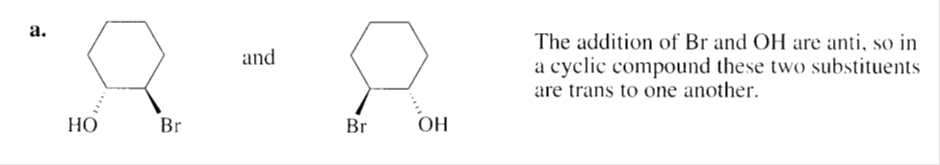
Chapter 6: Q47P (page 275)
a. What products will be obtained from the addition of Br2 to cyclohexene if H2O is added to the reaction
mixture?
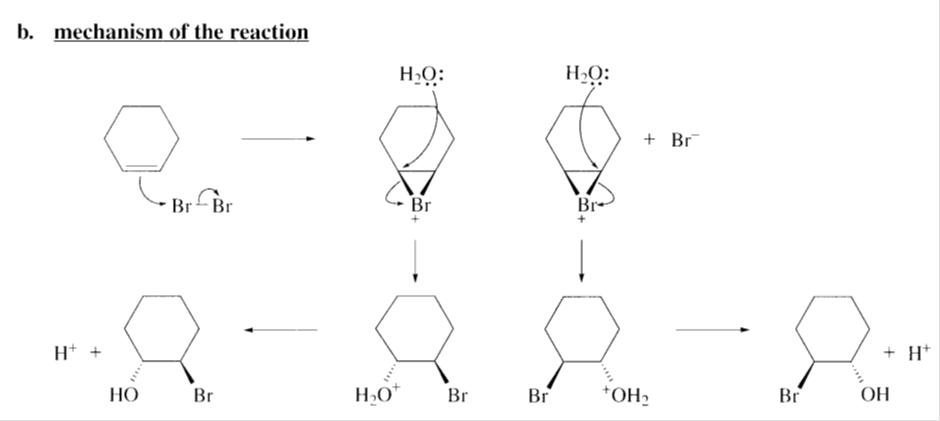
b. Propose a mechanism for the reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q47P (page 275)
a. What products will be obtained from the addition of Br2 to cyclohexene if H2O is added to the reaction
mixture?
b. Propose a mechanism for the reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
When fumarate reacts with D2O in the presence of the enzyme fumarase, only one isomer of the product is formed, as shown here. Is the enzyme catalyzing a syn or an anti-addition of D2O?
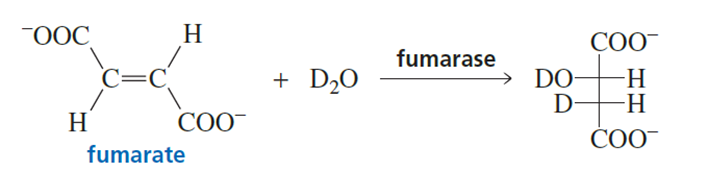
What stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2?
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
d. Is the reaction of 1-butene with HBr regioselective?
e. Is it stereoselective?
f. Is it stereospecific?
Draw the mechanism for the reaction of cyclohexene with HCl
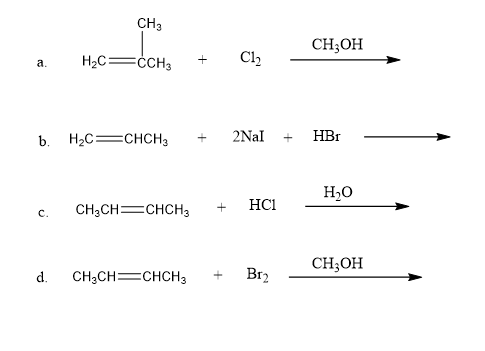
Each of the following reactions has two nucleophiles that could add to the intermediate formed by the reaction of the alkene with an electrophile. What is the major product of each reaction
What do you think about this solution?
We value your feedback to improve our textbook solutions.