Chapter 6: Q23P (page 256)
Each of the following reactions has two nucleophiles that could add to the intermediate formed by the reaction of the alkene with an electrophile. What is the major product of each reaction
Short Answer
Learning Materials
Features
Discover
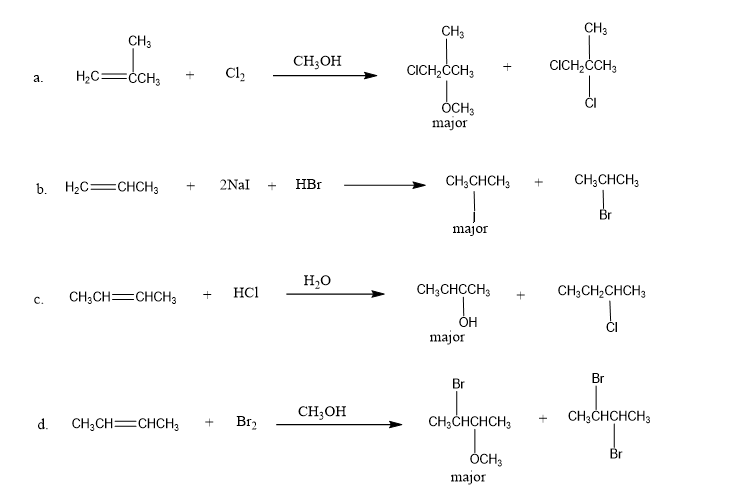
Chapter 6: Q23P (page 256)
Each of the following reactions has two nucleophiles that could add to the intermediate formed by the reaction of the alkene with an electrophile. What is the major product of each reaction


All the tools & learning materials you need for study success - in one app.
Get started for free
What stereoisomers are obtained from hydroboration–oxidation of the following compounds?Assign an Ror Sconfiguration to each asymmetric center.
a. cyclohexene c. cis-2-butene
b. 1-ethylcyclohexene d. (Z)-3,4-dimethyl-3-hexene
a. What alkene would give only a ketone with three carbons as a product of oxidative cleavage?
b. What alkenes would give only an aldehyde with four carbons as a product of oxidative cleavage?
a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl cation?
b. Which is more stable: a methyl cation or an ethyl cation? Why?
a. What is the major product obtained from the reaction of propene and Br2plus excess Cl-?
b. Indicate the relative amounts of the stereoisomers that are obtained.
What stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2?
What do you think about this solution?
We value your feedback to improve our textbook solutions.