Chapter 6: Q31P (page 262)
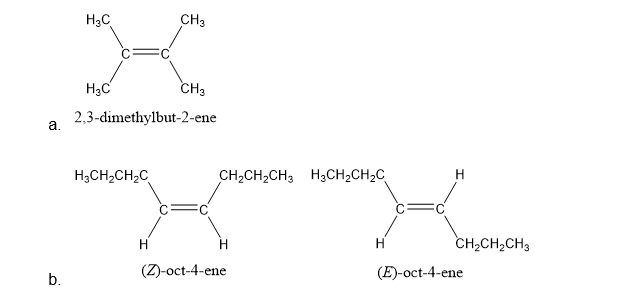
a. What alkene would give only a ketone with three carbons as a product of oxidative cleavage?
b. What alkenes would give only an aldehyde with four carbons as a product of oxidative cleavage?
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q31P (page 262)
a. What alkene would give only a ketone with three carbons as a product of oxidative cleavage?
b. What alkenes would give only an aldehyde with four carbons as a product of oxidative cleavage?

All the tools & learning materials you need for study success - in one app.
Get started for free
What stereoisomers would you expect to obtain from each of the following reactions

eDraw structures for the following:
a. 2-propyloxirane
b. cyclohexene oxide
c. 2,2,3,3-tetramethyloxirane
d. 2,3-epoxy-2-methylpentane
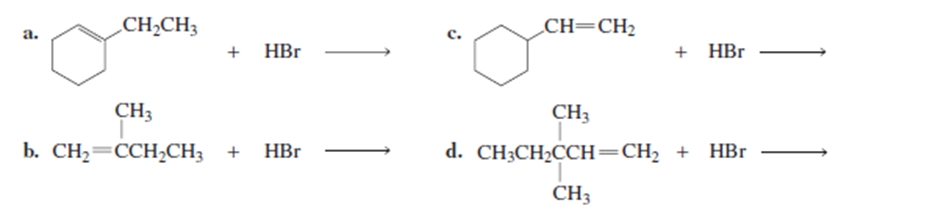
What is the major product of each of the following reactions?
Why are Na+ and K+ unable to form covalent bonds?
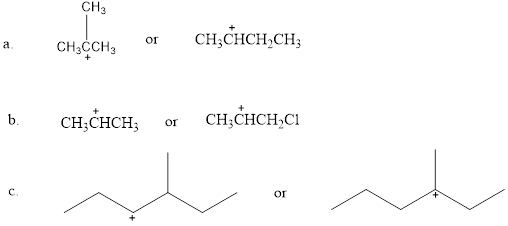
Which is more stable?
What do you think about this solution?
We value your feedback to improve our textbook solutions.