Chapter 6: Q48P (page 275)
What stereoisomers would you expect to obtain from each of the following reactions
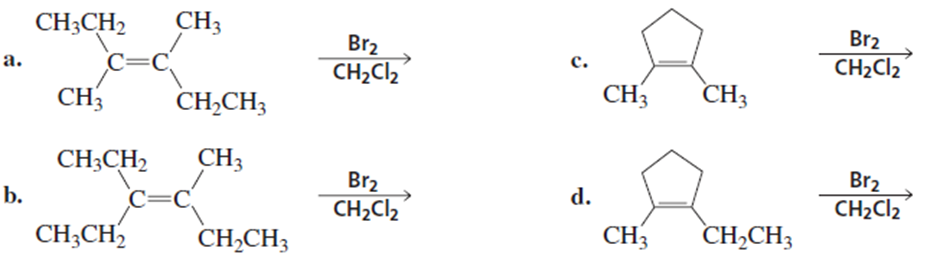
Short Answer
a)

b)
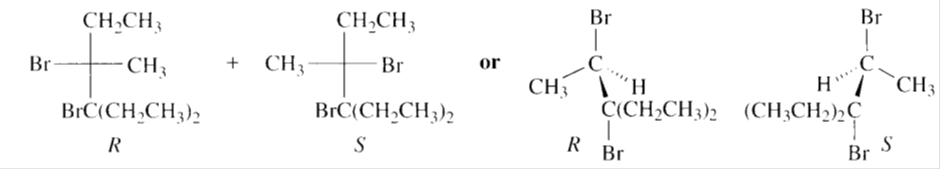
c)
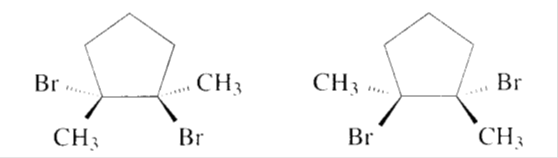
d)
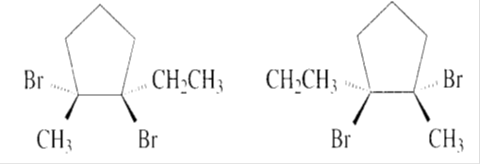
Learning Materials
Features
Discover
Chapter 6: Q48P (page 275)
What stereoisomers would you expect to obtain from each of the following reactions
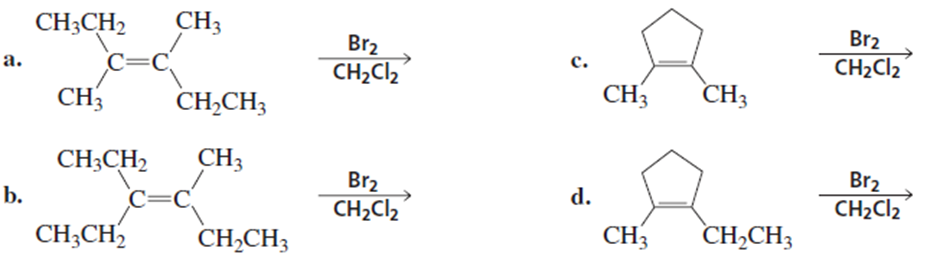
a)

b)
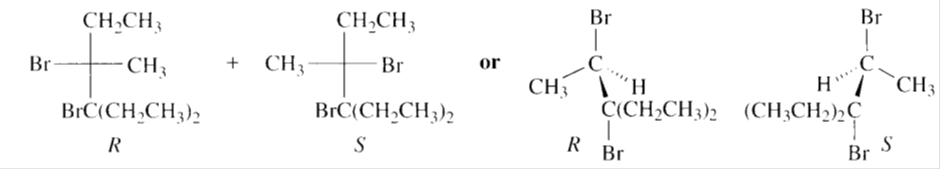
c)
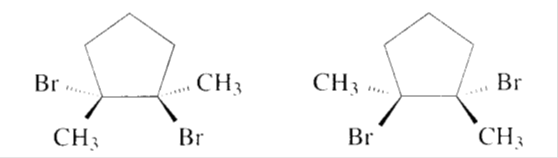
d)
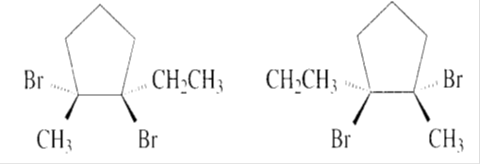
All the tools & learning materials you need for study success - in one app.
Get started for free
a. What is the product of the reaction of fumarate and H2O when H2SO4 is used as a catalyst instead of fumarase?
b. What is the product of the reaction of maleate and H2O when H2SO4 is used as a catalyst instead of fumarase?
Which electrophilic addition reactions
a. form a carbocation intermediate? c. form a three-membered ring intermediate?
b. form no intermediate? d. form a five-membered ring intermediate?
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
d. Is the reaction of 1-butene with HBr regioselective?
e. Is it stereoselective?
f. Is it stereospecific?
Draw the mechanism for the reaction of cyclohexene with HCl
Propose a mechanism for the following reaction:
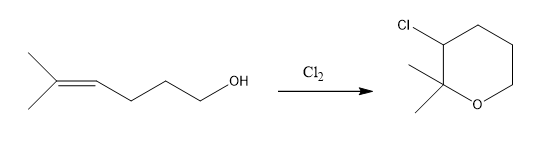
What do you think about this solution?
We value your feedback to improve our textbook solutions.