Chapter 6: Q1P (page 235)
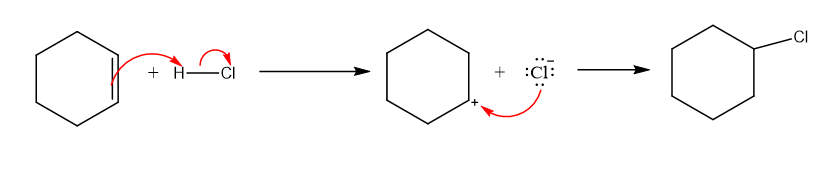
Draw the mechanism for the reaction of cyclohexene with HCl
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q1P (page 235)
Draw the mechanism for the reaction of cyclohexene with HCl

All the tools & learning materials you need for study success - in one app.
Get started for free
a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl cation?
b. Which is more stable: a methyl cation or an ethyl cation? Why?
a. Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.
b. Would both alkenes form the same alkyl halide if DBr were used instead of HBr? (D is an isotope of H, so D+ reacts like H+.)
eDraw structures for the following:
a. 2-propyloxirane
b. cyclohexene oxide
c. 2,2,3,3-tetramethyloxirane
d. 2,3-epoxy-2-methylpentane
What hydrocarbon forms the following products after reaction first with ozone and then with dimethyl sulfide?
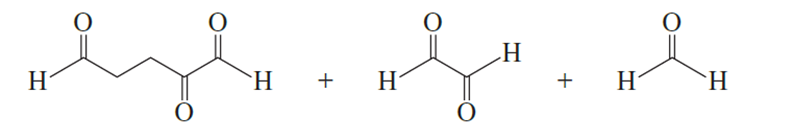
Ozonolysis of an alkene, followed by treatment with dimethyl sulfide, forms the following product(s). Identify the alkene in each case.

What do you think about this solution?
We value your feedback to improve our textbook solutions.