Chapter 6: Q100P (page 287)
What stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2?
Short Answer
The formation of the stereoisomer is trans-1,2dichloro-(S)-3-methyl- pentane.
Learning Materials
Features
Discover
Chapter 6: Q100P (page 287)
What stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2?
The formation of the stereoisomer is trans-1,2dichloro-(S)-3-methyl- pentane.
All the tools & learning materials you need for study success - in one app.
Get started for free
What reagents are needed to carry out the following syntheses?
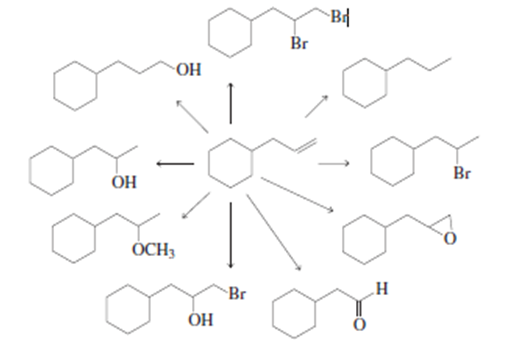
a. What products will be obtained from the addition of Br2 to cyclohexene if H2O is added to the reaction
mixture?
b. Propose a mechanism for the reaction.
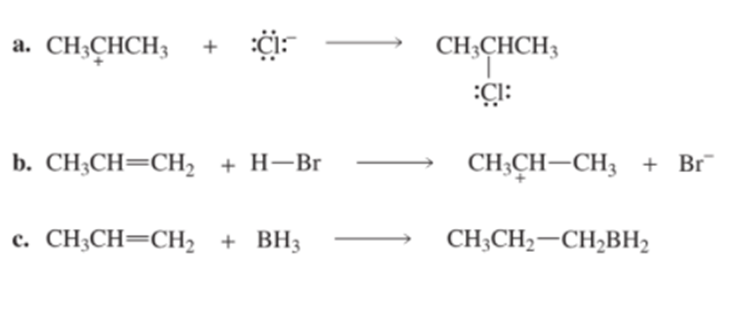
Identify the electrophile and the nucleophile in each of the following reaction steps, and then draw curved arrows to illustrate the bond-making and bond-breaking processes.
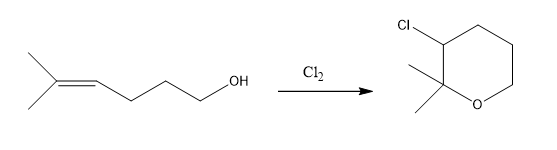
Propose a mechanism for the following reaction:
eDraw structures for the following:
a. 2-propyloxirane
b. cyclohexene oxide
c. 2,2,3,3-tetramethyloxirane
d. 2,3-epoxy-2-methylpentane
What do you think about this solution?
We value your feedback to improve our textbook solutions.