Chapter 6: Q101P (page 287)
Propose a mechanism for the following reaction:
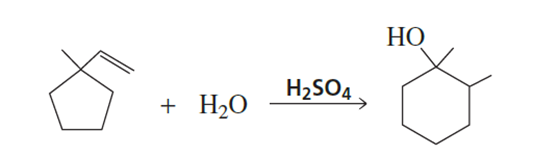
Short Answer
The reaction mechanism is an acid hydrolysis reaction and it is given below.
Learning Materials
Features
Discover
Chapter 6: Q101P (page 287)
Propose a mechanism for the following reaction:
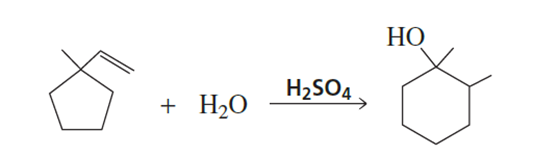
The reaction mechanism is an acid hydrolysis reaction and it is given below.
All the tools & learning materials you need for study success - in one app.
Get started for free
When Br2adds to a cis alkene that has different substituents attached to each of the two sp2carbons, such as cis-2-heptene, identical amounts of the two threo enantiomers are obtained even though Br-is more likely to attack the less sterically hindered carbon of the bromonium ion. Explain why identical amounts of the two enantiomers are obtained.
What will be the product of the preceding reaction if HBr is used in place of Br2?
What will be the major product of the reaction of 2-methyl-2-butene with each of the following reagents?
a. HBr
b. HI
c. Cl2/CH2Cl2
d. O3, −78 °C, followed by (CH3)2S
e. H2/Pd
f. MCPBA (a peroxyacid)
g. H2O + H2SO4
h. Br2/CH2Cl2
i. Br2/H2O
j. Br2/CH3OH
k. BH3/THF, followed by H2O2, HO-, H2O
a. Is the reaction of 2-butene with HBr regioselective?
b. Is it stereoselective?
c. Is it stereospecific?
d. Is the reaction of 1-butene with HBr regioselective?
e. Is it stereoselective?
f. Is it stereospecific?
Why are Na+ and K+ unable to form covalent bonds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.