Chapter 6: Q57P (page 282)
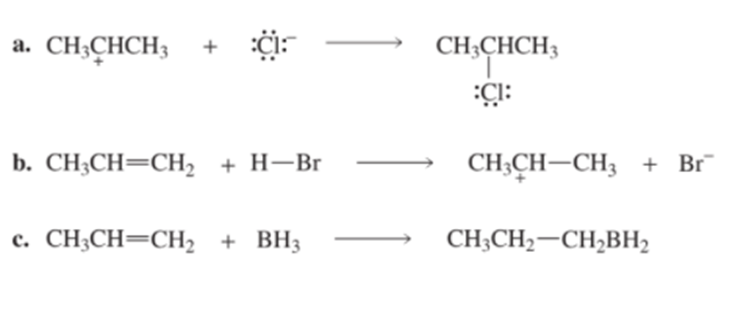
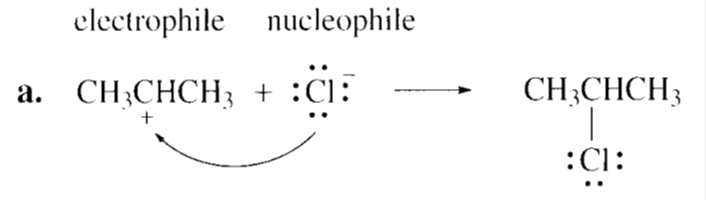
Identify the electrophile and the nucleophile in each of the following reaction steps, and then draw curved arrows to illustrate the bond-making and bond-breaking processes.
Short Answer

Learning Materials
Features
Discover
Chapter 6: Q57P (page 282)
Identify the electrophile and the nucleophile in each of the following reaction steps, and then draw curved arrows to illustrate the bond-making and bond-breaking processes.



All the tools & learning materials you need for study success - in one app.
Get started for free
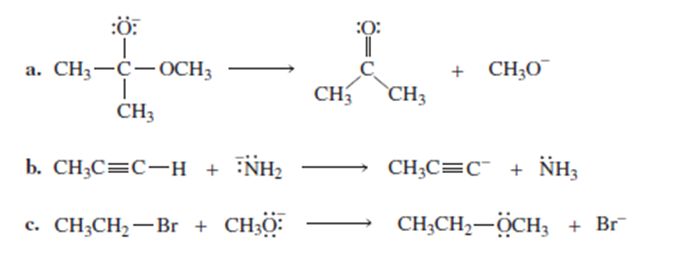
Draw curved arrows to show the flow of electrons responsible for the conversion of the following reactants into products.
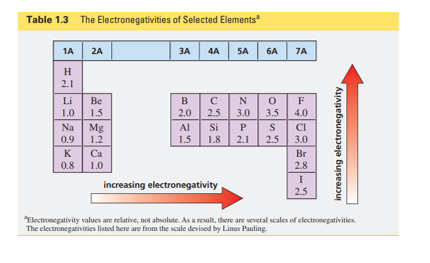
What is the product of the addition of I-Cl to 1-butene? (Hint: Chlorine is more electronegative than iodine [Table 1.3].)
When Br2adds to a cis alkene that has different substituents attached to each of the two sp2carbons, such as cis-2-heptene, identical amounts of the two threo enantiomers are obtained even though Br-is more likely to attack the less sterically hindered carbon of the bromonium ion. Explain why identical amounts of the two enantiomers are obtained.
Propose a mechanism for the following reaction:
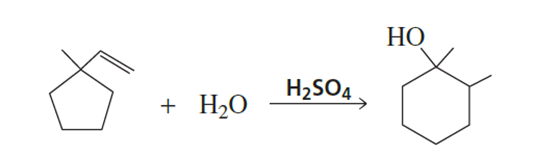
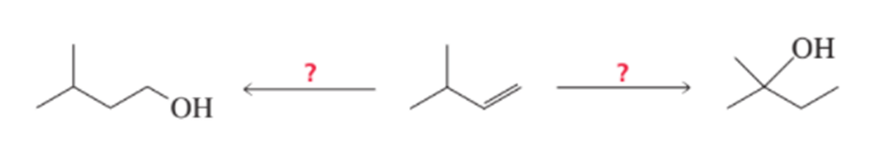
What reagents are needed to synthesize the following alcohols?
What do you think about this solution?
We value your feedback to improve our textbook solutions.