Chapter 6: Q60P (page 283)
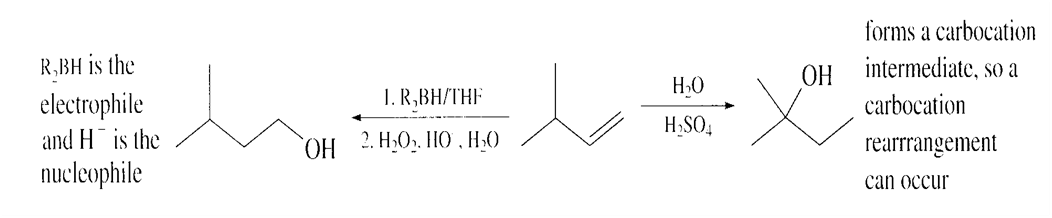
What reagents are needed to synthesize the following alcohols?
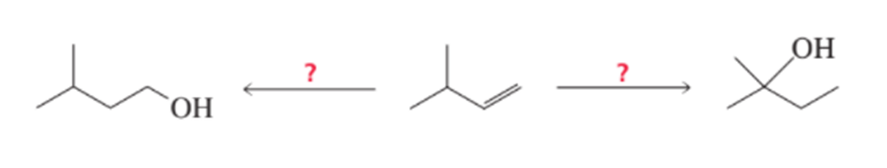
Short Answer
Here we have to find What reagents are needed to synthesize the following alcohols:
Learning Materials
Features
Discover
Chapter 6: Q60P (page 283)
What reagents are needed to synthesize the following alcohols?

Here we have to find What reagents are needed to synthesize the following alcohols:

All the tools & learning materials you need for study success - in one app.
Get started for free
:a. How does the first step in the reaction of propene with Br2 differ from the first step in the reaction of propene with HBr?
b. To understand why Br- adds to a carbon of the bromonium ion rather than to the positively charged bromine, draw the product that would be obtained if Br- did add to bromine.
What stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2?
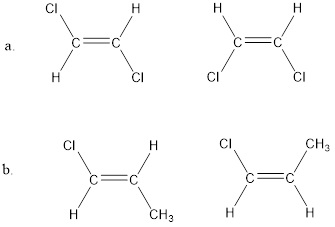
Which compound has the greater dipole moment?
The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.
Which electrophilic addition reactions
a. form a carbocation intermediate? c. form a three-membered ring intermediate?
b. form no intermediate? d. form a five-membered ring intermediate?
What do you think about this solution?
We value your feedback to improve our textbook solutions.