Chapter 6: Q63P (page 283)
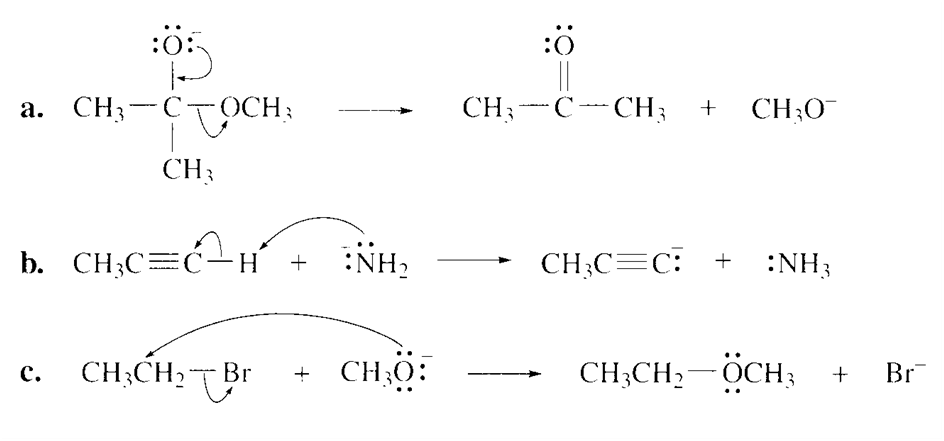
Draw curved arrows to show the flow of electrons responsible for the conversion of the following reactants into products.
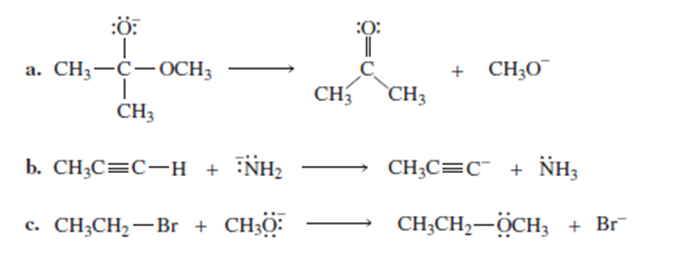
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q63P (page 283)
Draw curved arrows to show the flow of electrons responsible for the conversion of the following reactants into products.


All the tools & learning materials you need for study success - in one app.
Get started for free
What stereoisomers are obtained from the following reactions?
a. trans-2-butene + HBr
b. (Z)-3-methyl-2-pentene + HBr
c. (E)-3-methyl-2-pentene + HBr
d. cis-3-hexene + HBr
e. cis-2-pentene + Br2
f. 1-hexene + Br2
a. What is the major product obtained from the reaction of propene and Br2plus excess Cl-?
b. Indicate the relative amounts of the stereoisomers that are obtained.
When 3-methyl-1-butene reacts with HBr, two alkyl halides are formed: 2-bromo-3-methylbutane and 2-bromo-2-methylbutane. Propose a mechanism that explains the formation of these two products.
What reagents are needed to carry out the following syntheses?
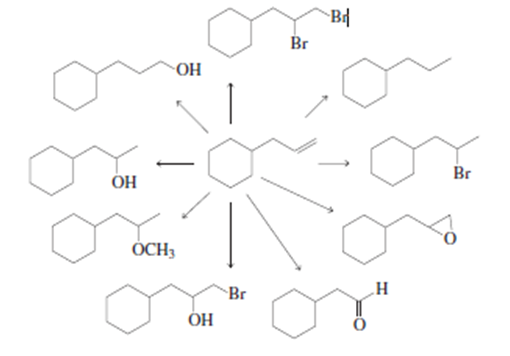
Why are Na+ and K+ unable to form covalent bonds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.