Chapter 6: Q62P (page 283)
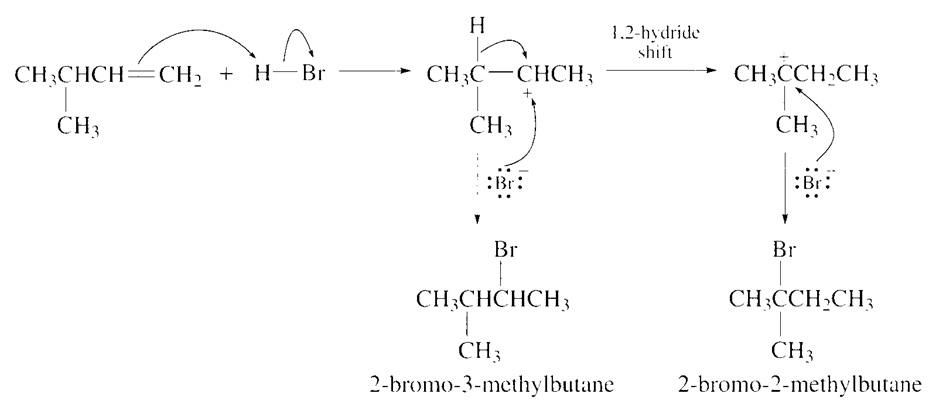
When 3-methyl-1-butene reacts with HBr, two alkyl halides are formed: 2-bromo-3-methylbutane and 2-bromo-2-methylbutane. Propose a mechanism that explains the formation of these two products.
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q62P (page 283)
When 3-methyl-1-butene reacts with HBr, two alkyl halides are formed: 2-bromo-3-methylbutane and 2-bromo-2-methylbutane. Propose a mechanism that explains the formation of these two products.

All the tools & learning materials you need for study success - in one app.
Get started for free
What aspect of the structure of the alkene does ozonolysis not tell you ?
What stereoisomers would you expect to obtain from each of the following reactions
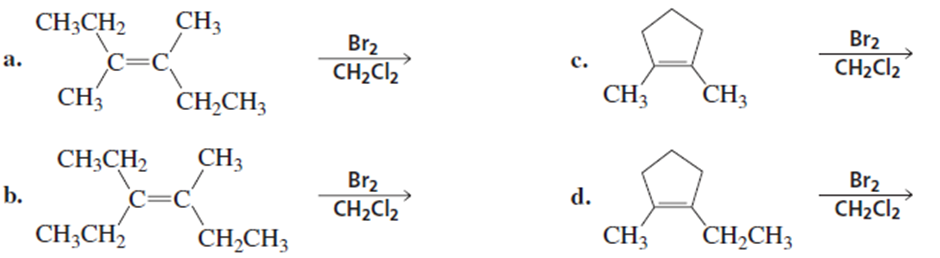
Which electrophilic addition reactions
a. form a carbocation intermediate? c. form a three-membered ring intermediate?
b. form no intermediate? d. form a five-membered ring intermediate?
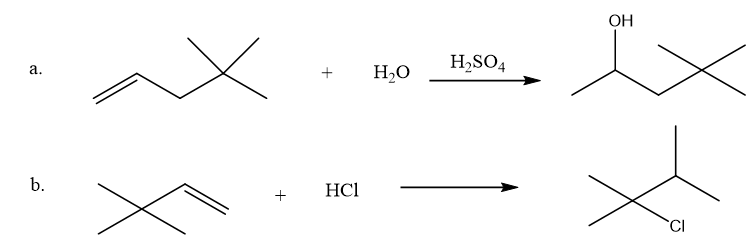
Propose a mechanism for each of the following reactions:
a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl cation?
b. Which is more stable: a methyl cation or an ethyl cation? Why?
What do you think about this solution?
We value your feedback to improve our textbook solutions.