Chapter 15: Q33P (page 712)
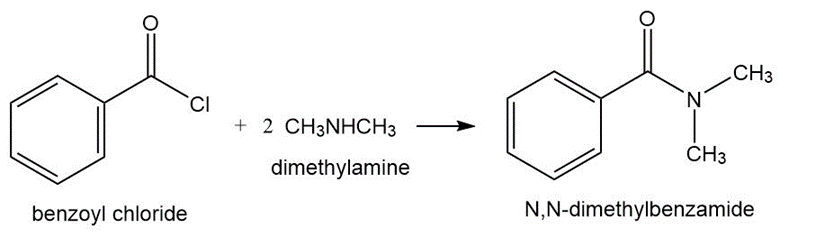
Problem:What acyl chloride and amine are required to synthesize the following amides?
a. N-ethylbutanamide b. N,N-dimethylbenzamide
Short Answer
Answer
a.

b.
Learning Materials
Features
Discover
Chapter 15: Q33P (page 712)
Problem:What acyl chloride and amine are required to synthesize the following amides?
a. N-ethylbutanamide b. N,N-dimethylbenzamide
Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Is the acid-catalyzed hydrolysis of acetamide a reversible or an irreversible reaction? Explain.
Write the mechanism for the acid-catalyzed reaction of an amide with an alcohol to form an ester.
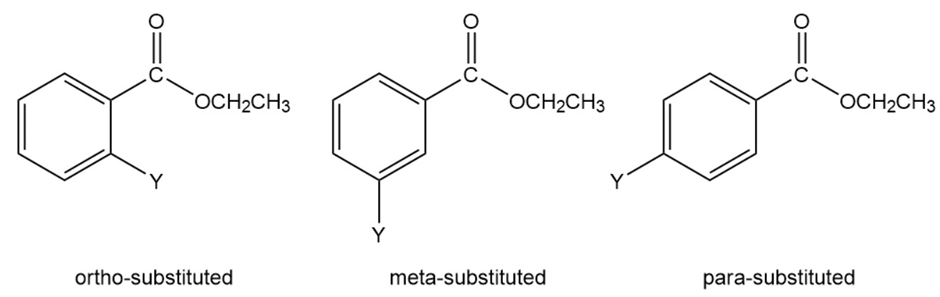
Information about the mechanism of the reaction undergone by a series of substituted benzenes can be obtained by plotting logarithm of the observed rate constant determined at particular pH against the Hammett substituent constant for the particular substituent. The value for hydrogen is zero. Electron-donating substituents have negative values; the more strongly electron withdrawing the substituent, the more positiveits s value. The slope of a plot of the logarithm of the rate constant versusis called the (rho) value. The value for the hydroxide-ion-promoted hydrolysis of a series of meta- and para-substituted ethyl
benzoates is ; the value for amide formation for the reaction of a series of meta- and para-substituted anilines with benzoyl chloride is .
a. Propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride.
b. How does acetic anhydride make it easier to form the anhydride?
What is the product of an acyl substitution reaction—a new carboxylic acid derivative, a mixture of two carboxylic acid derivatives, or no reaction—if the new group in the tetrahedral intermediate is the following?
a. a stronger base than the substituent that is attached to the acyl group
b. a weaker base than the substituent that is attached to the acyl group
c. similar in basicity to the substituent that is attached to the acyl group
What do you think about this solution?
We value your feedback to improve our textbook solutions.