Chapter 15: Q32P (page 712)
Problem:Propose a mechanism for the following reaction. (Hint: Number the carbons to help you see where they end up in the product.)
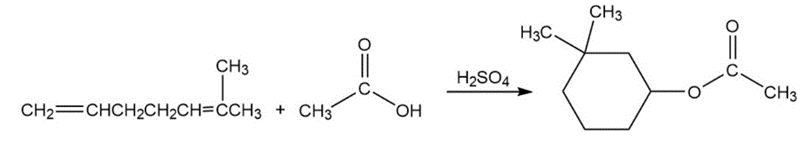
Short Answer
Answer
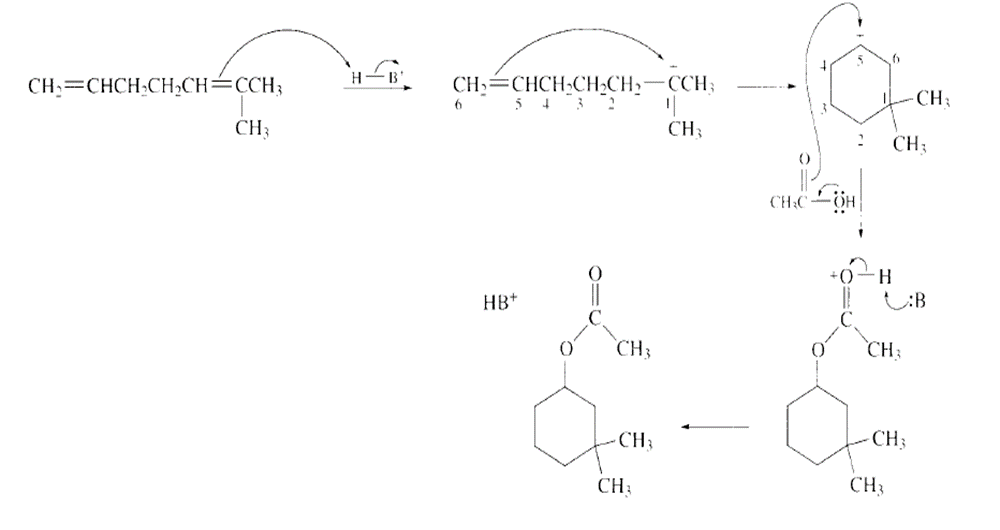
Learning Materials
Features
Discover
Chapter 15: Q32P (page 712)
Problem:Propose a mechanism for the following reaction. (Hint: Number the carbons to help you see where they end up in the product.)

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
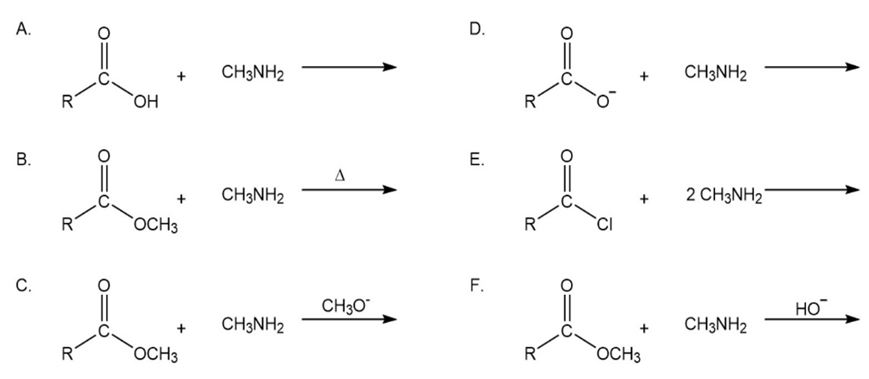
Problem: Which of the following reactions lead to the Formation of an amide?
Question: What product do you expect to obtain from each of the following reactions?
a.
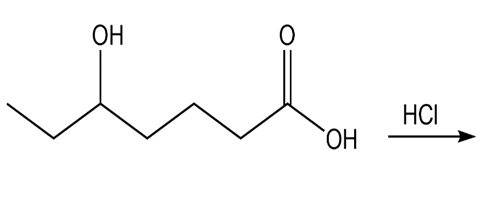
b.
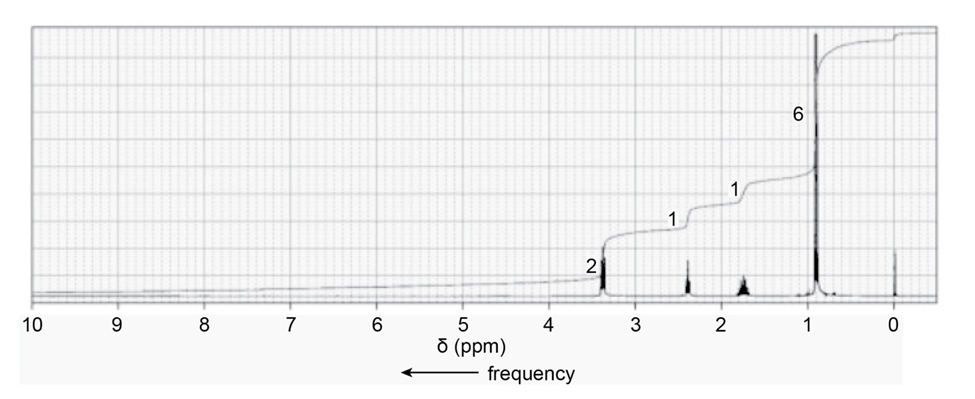
When a compound with molecular formula undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following NMR spectrum. Identify the compound.
The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, does not work with primary alcohols.
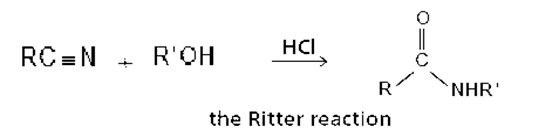
a. Propose a mechanism for the Ritter reaction.
b. Why does the Ritter reaction not work with primary alcohols?
c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?
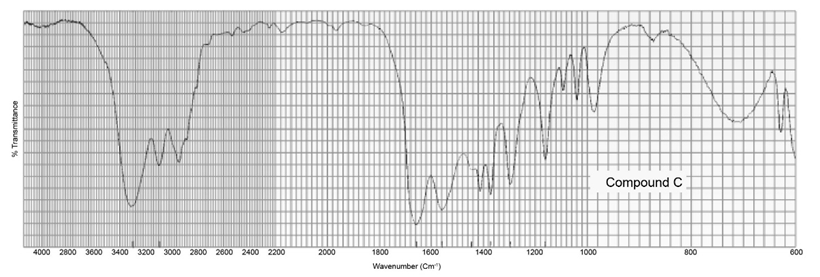
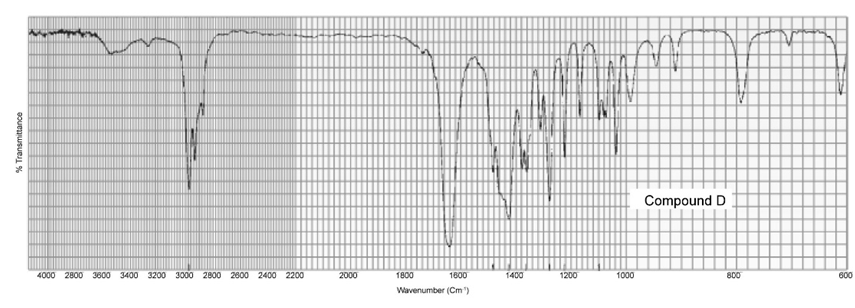
Two products, A and B, are obtained from the reaction of 1-bromobutane with NH3 . Compound A reacts with acetyl chloride to form C, and compound B reacts with acetyl chloride to form D. The IR spectra of C and D are shown. Identify A, B, C, and D.
What do you think about this solution?
We value your feedback to improve our textbook solutions.