Chapter 15: Q87P (page 737)
Question: What product do you expect to obtain from each of the following reactions?
a.
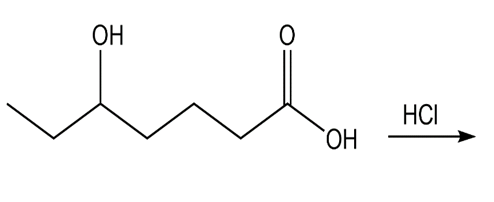
b.
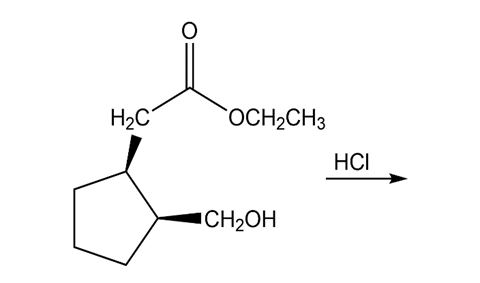
Short Answer
Answer
a.
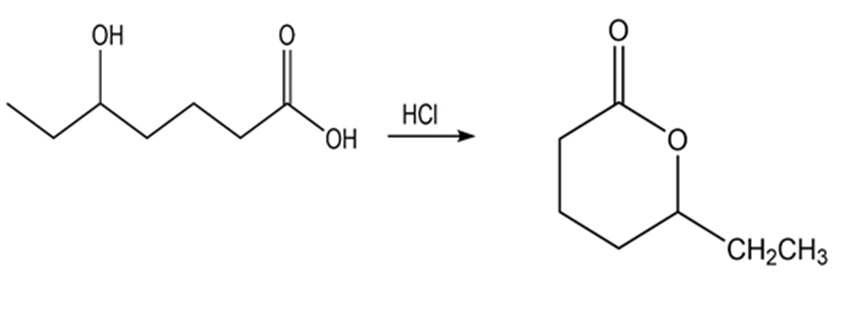
b.
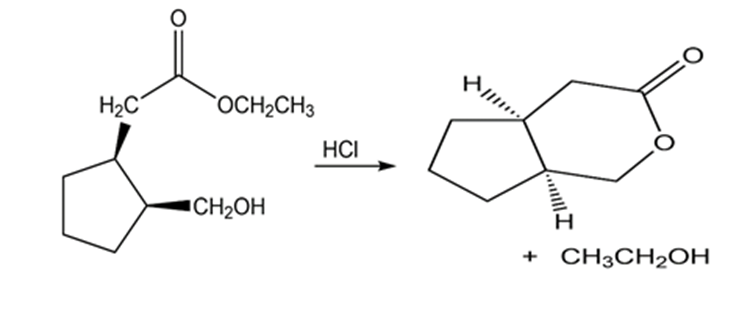
Learning Materials
Features
Discover
Chapter 15: Q87P (page 737)
Question: What product do you expect to obtain from each of the following reactions?
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Which alkyl halides form the carboxylic acids listed hereafter reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
a. butyric acid
b. isovaleric acid
c. cyclohexane carboxylic acid
Question: Draw a structure for each of the following:
a. N,N-dimethylhexanamide
b. 3,3-dimethylhexanamide
c. Cyclohexanecarbonyl chloride
d. Propanenitrile
e. propionamide
f. sodium acetate
g. benzoic anhydride
h. b-valerolactone
i. 3-methylbutanenitrile
j. cycloheptanecarboxylic acid
k. benzoyl chloride
Primary amines can also be prepared by the reaction of an alkyl halide with azide ion, followed by catalytic hydrogenation. What advantage do this method and the Gabriel synthesis have over the synthesis of a primary amine using an alkyl halide and ammonia?
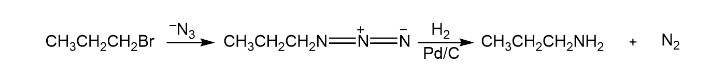
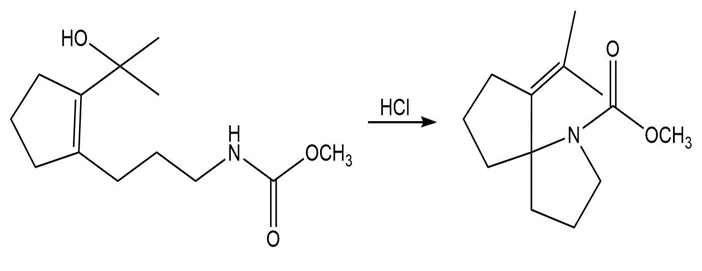
Propose a mechanism that accounts for the formation of the product.
Problem:Show how each of the following esters could be prepared using a carboxylic acid as one of the startingmaterials:
a. methyl butyrate (odor of apples) b. octyl acetate (odor of oranges)
Problem:Show how each of the following esters could be prepared using a carboxylic acid as one of the startingmaterials:
a. methyl butyrate (odor of apples) b. octyl acetate (odor of oranges)
What do you think about this solution?
We value your feedback to improve our textbook solutions.