Chapter 15: Q90P (page 738)
Propose a mechanism that accounts for the formation of the product.
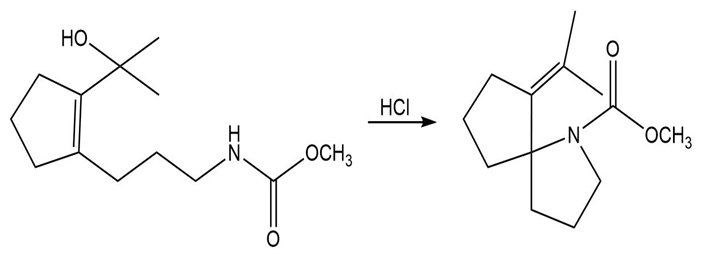
Short Answer
The proposed mechanism is shown below.

Learning Materials
Features
Discover
Chapter 15: Q90P (page 738)
Propose a mechanism that accounts for the formation of the product.

The proposed mechanism is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
When butanoic acid and 18O-labelled methanol react under acidic conditions, what compounds are labelled when the reaction has reached equilibrium?
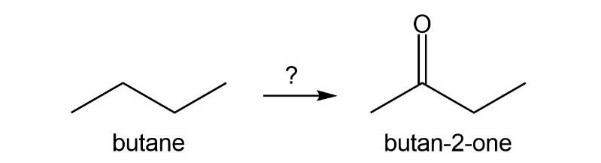
Describe how the target molecule (butanone) can be synthesized in a high yield from butane.
Which alkyl halides form the carboxylic acids listed hereafter reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
a. butyric acid
b. isovaleric acid
c. cyclohexane carboxylic acid
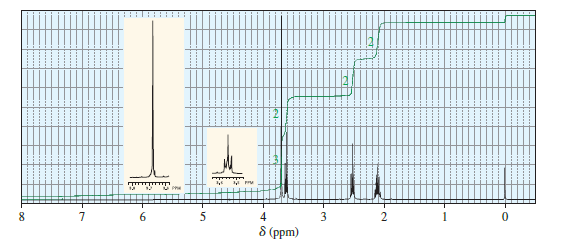
When treated with an equivalent of methanol, compound A, with the molecular formula C4H6Cl2O, forms the compound whose 1H NMR spectrum is shown here. Identify compound A.
Question: Draw a structure for each of the following:
a. N,N-dimethylhexanamide
b. 3,3-dimethylhexanamide
c. Cyclohexanecarbonyl chloride
d. Propanenitrile
e. propionamide
f. sodium acetate
g. benzoic anhydride
h. b-valerolactone
i. 3-methylbutanenitrile
j. cycloheptanecarboxylic acid
k. benzoyl chloride
What do you think about this solution?
We value your feedback to improve our textbook solutions.