Chapter 15: Q73P (page 735)
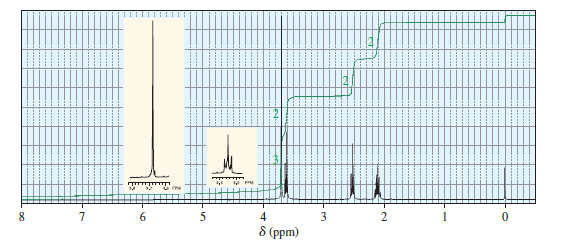
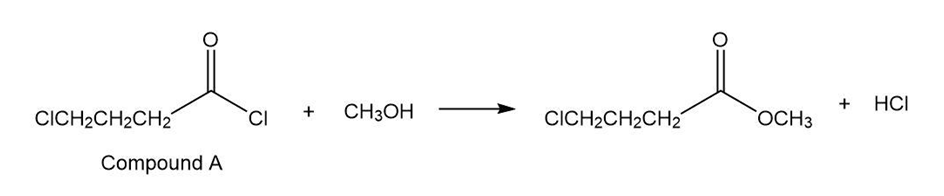
When treated with an equivalent of methanol, compound A, with the molecular formula C4H6Cl2O, forms the compound whose 1H NMR spectrum is shown here. Identify compound A.
Short Answer
Mechanism followed as:
Learning Materials
Features
Discover
Chapter 15: Q73P (page 735)
When treated with an equivalent of methanol, compound A, with the molecular formula C4H6Cl2O, forms the compound whose 1H NMR spectrum is shown here. Identify compound A.

Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
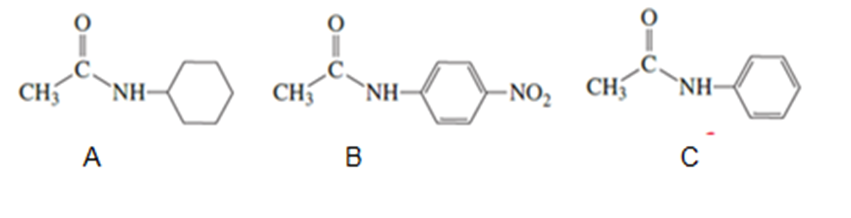
Rank the following amides from greatest reactivity to least reactivity toward acid-catalyzed hydrolysis:
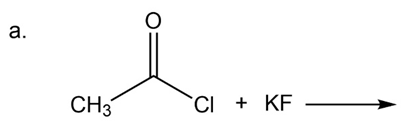
Propose a mechanism for the reaction of an acyl chloride with acetate ion to form an acid anhydride
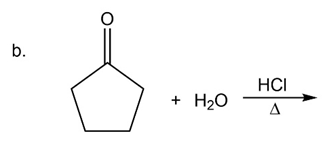
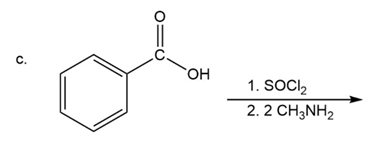
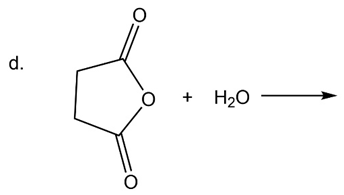
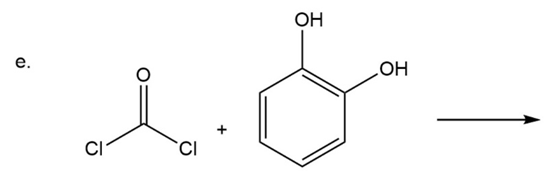
What are the products of the following reactions?
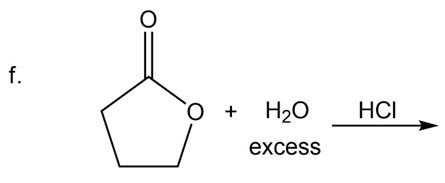
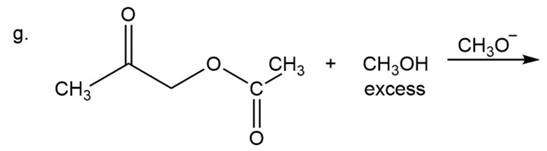
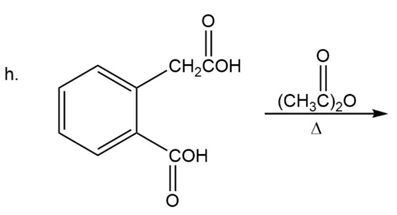
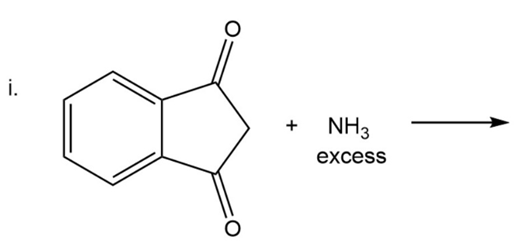
Which is a correct statement?
A. The delocalization energy of an ester is about 18 kcal/mol, and the delocalization energy of an amide is about 10 kcal/mol.
B. The delocalization energy of an ester is about 10 kcal/mol, and the delocalization energy of an amide is about 18 kcal/mol.
a. Propose a mechanism for the reaction of acetic anhydride with water.
b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.