Chapter 15: Q72P (page 735)
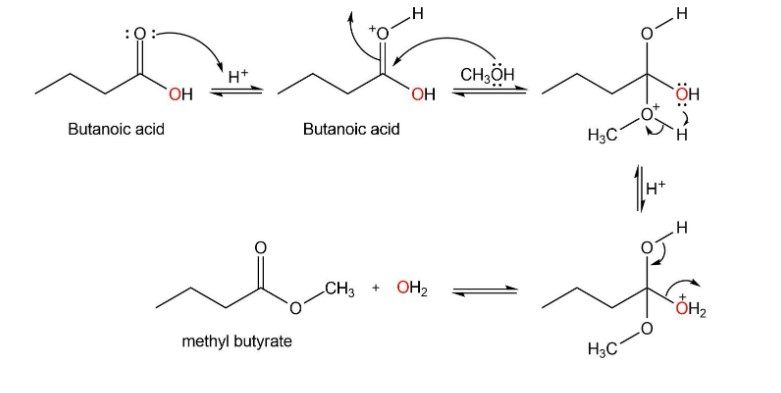
When butanoic acid and 18O-labelled methanol react under acidic conditions, what compounds are labelled when the reaction has reached equilibrium?
Short Answer
Mechanism followed as:
Learning Materials
Features
Discover
Chapter 15: Q72P (page 735)
When butanoic acid and 18O-labelled methanol react under acidic conditions, what compounds are labelled when the reaction has reached equilibrium?
Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
a.Identify the two products obtained from the following reaction:
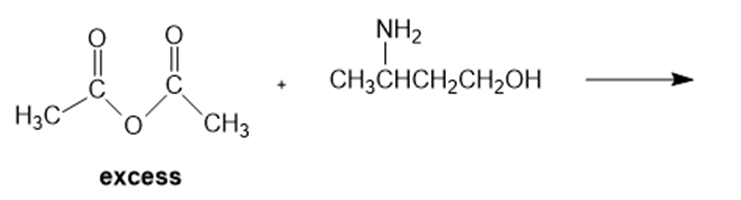
b. A student carried out the preceding reaction, but stopped it before it was half over, whereupon he isolated the major product. He was surprised to find
that the product he isolated was neither of the products obtained when the reaction was allowed to go to completion. What product did he isolate?
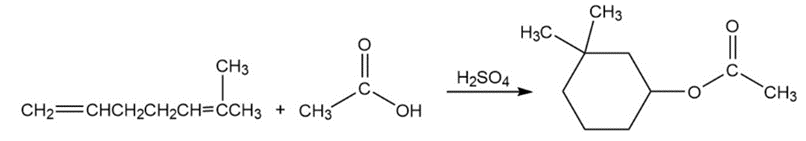
Problem:Propose a mechanism for the following reaction. (Hint: Number the carbons to help you see where they end up in the product.)
When a student treated butane dioic acid with thionyl chloride, she was surprised to find that the product she obtained was an anhydride rather than an acyl chloride. Propose a mechanism to explain why she obtained an anhydride.
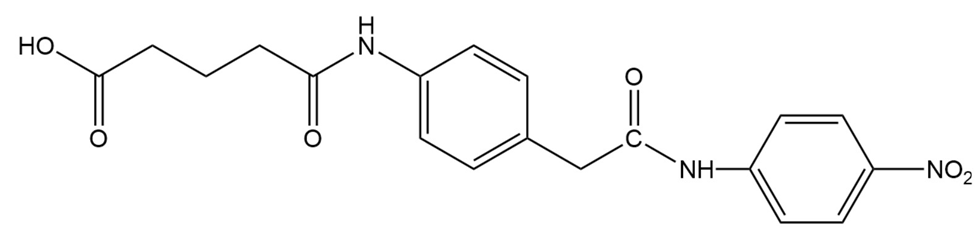
Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of antibody is carried out in the presence of a transition state analog-a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of structurally similar ester:

a. Draw a possible transition state for the hydrolysis of the reaction.
b. The following transition state analog is used to generate a catalytic antibody for the catalysis of ester hydrolysis. Draw the structure of an ester whose rate of hydrolysis would be increased by this catalytic antibody.

c. Design a transition state analog that catalyzes amide hydrolysis at the amide group indicated.
When treated with an equivalent of methanol, compound A, with the molecular formula C4H6Cl2O, forms the compound whose 1H NMR spectrum is shown here. Identify compound A.
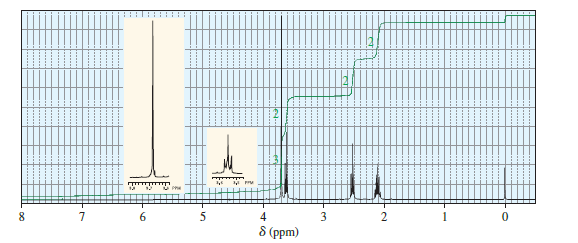
What do you think about this solution?
We value your feedback to improve our textbook solutions.