Chapter 15: Q44P (page 721)
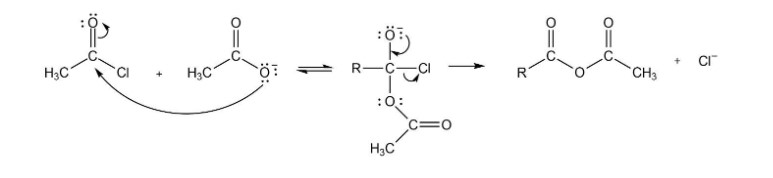
Propose a mechanism for the reaction of an acyl chloride with acetate ion to form an acid anhydride
Short Answer
Mechanism followed as:
Learning Materials
Features
Discover
Chapter 15: Q44P (page 721)
Propose a mechanism for the reaction of an acyl chloride with acetate ion to form an acid anhydride
Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
Which alkyl halides form the carboxylic acids listed hereafter reaction with sodium cyanide followed by heating the product in an acidic aqueous solution?
a. butyric acid
b. isovaleric acid
c. cyclohexane carboxylic acid
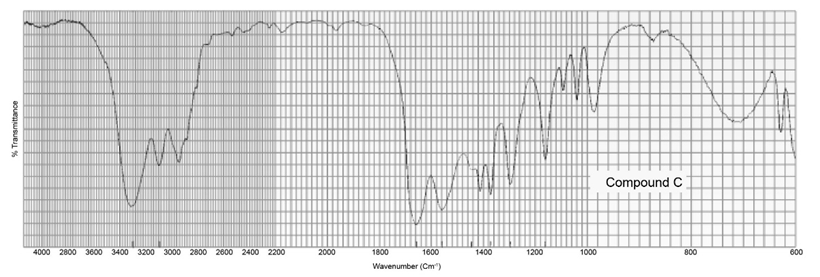
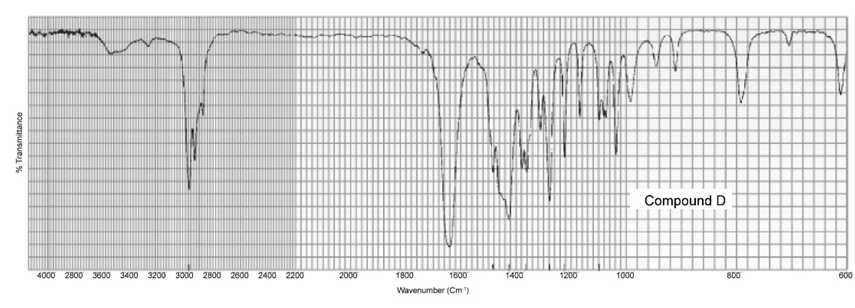
Two products, A and B, are obtained from the reaction of 1-bromobutane with NH3 . Compound A reacts with acetyl chloride to form C, and compound B reacts with acetyl chloride to form D. The IR spectra of C and D are shown. Identify A, B, C, and D.
What alkyl bromide would you use in a Gabriel synthesis to prepare each of the following amines?
Pentylamine
Iso hexyl amine
Benzylamine
Cyclohexylamine
Name the following.
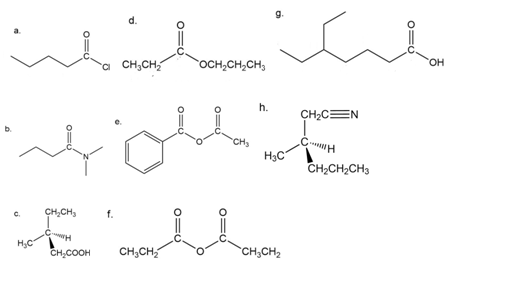
How could you use 1H NMR spectroscopy to distinguish the following esters?
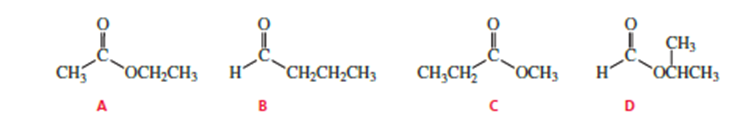
What do you think about this solution?
We value your feedback to improve our textbook solutions.