Chapter 15: Q43P (page 721)
a. Propose a mechanism for the reaction of acetic anhydride with water.
b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?
Short Answer
Mechanism followed as:

Learning Materials
Features
Discover
Chapter 15: Q43P (page 721)
a. Propose a mechanism for the reaction of acetic anhydride with water.
b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?
Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
Problem:D. N. Kursanov, a Russian chemist, proved that the bond that is broken in the hydroxide-ion-promoted hydrolysis of an ester is the acyl C-O bond, rather than the alkyl C-O bond, by studying the hydrolysis
of the following ester under basic conditions:
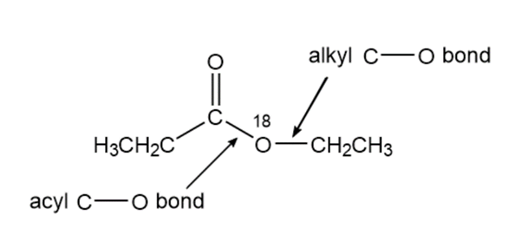
a. What products contained the label?
b. What product would have contained the label if the alkyl C-O bond had broken?
a. What is the product of the reaction of acetyl chloride with? Theof HCl is -7; theofis 15.7.
b. What is the product of the reaction of acetamide with? Theofis 36; theofis 15.7.
The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, does not work with primary alcohols.
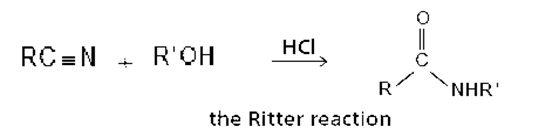
a. Propose a mechanism for the Ritter reaction.
b. Why does the Ritter reaction not work with primary alcohols?
c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?
If propionyl chloride is added to one equivalent of methylamine, only a 50% yield of N-methylpropanamide is obtained. If, however, the acyl chloride is added to two equivalents of methylamine, the yield of N-methylpropanamide is almost 100%. Explain these observations.
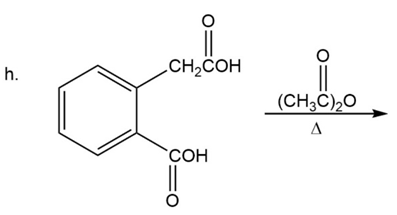
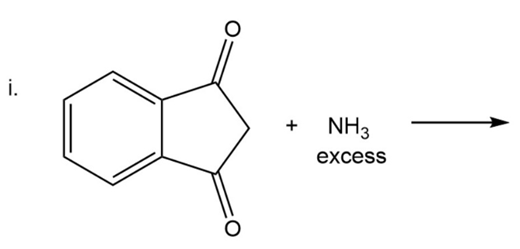
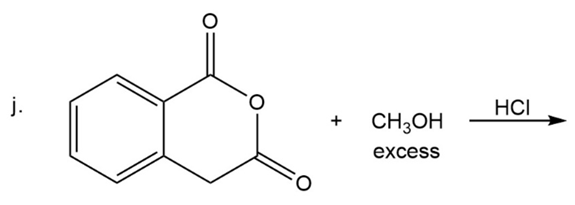
What are the products of the following reactions?
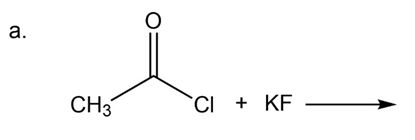
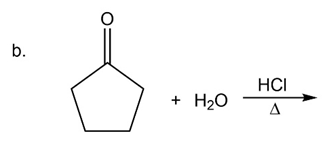
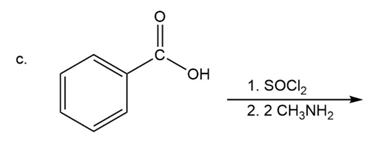
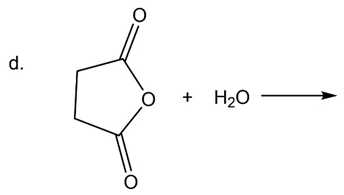

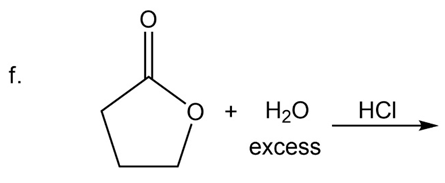
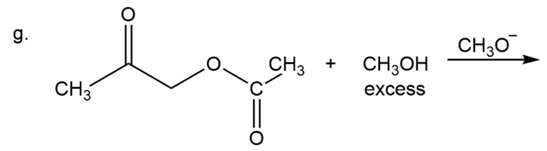
What do you think about this solution?
We value your feedback to improve our textbook solutions.