Information about the mechanism of the reaction undergone by a series of substituted benzenes can be obtained by plotting logarithm of the observed rate constant determined at particular pH against the Hammett substituent constant for the particular substituent. The value for hydrogen is zero. Electron-donating substituents have negative values; the more strongly electron withdrawing the substituent, the more positiveits s value. The slope of a plot of the logarithm of the rate constant versusis called the (rho) value. The value for the hydroxide-ion-promoted hydrolysis of a series of meta- and para-substituted ethyl
benzoates is ; the value for amide formation for the reaction of a series of meta- and para-substituted anilines with benzoyl chloride is .
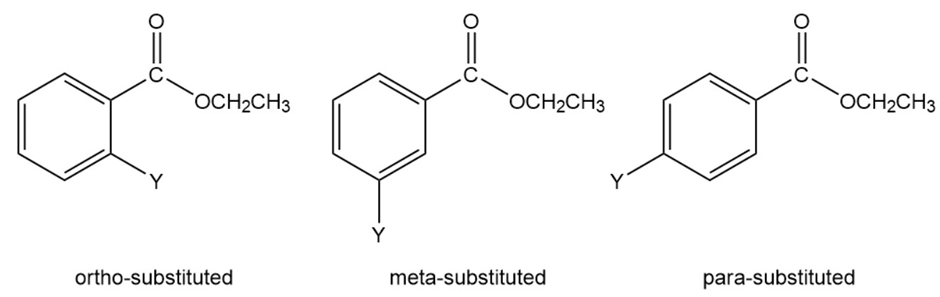
- Why doesone set of experiments give a positive role="math" localid="1652525783964" value, whereas the other set of experiments gives a negative value?
- Why were ortho-substituted compounds not included in the experiment?
- What do you predict the sign of the value to be for the ionization of a series of meta- and para-substituted benzoic acids?






