Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of antibody is carried out in the presence of a transition state analog-a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of structurally similar ester:
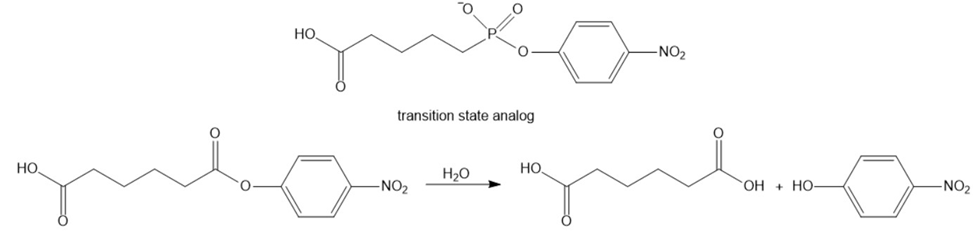
a. Draw a possible transition state for the hydrolysis of the reaction.
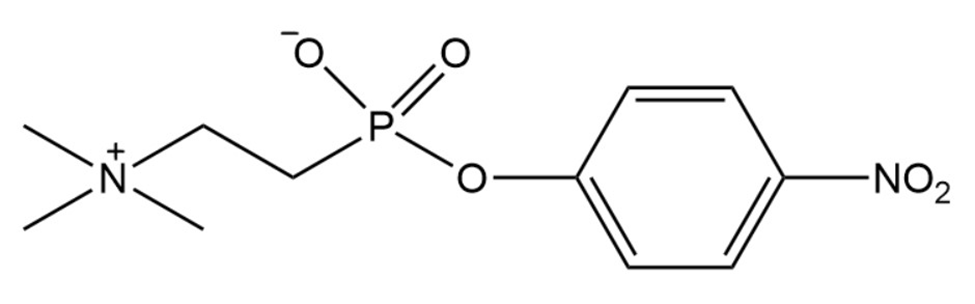
b. The following transition state analog is used to generate a catalytic antibody for the catalysis of ester hydrolysis. Draw the structure of an ester whose rate of hydrolysis would be increased by this catalytic antibody.
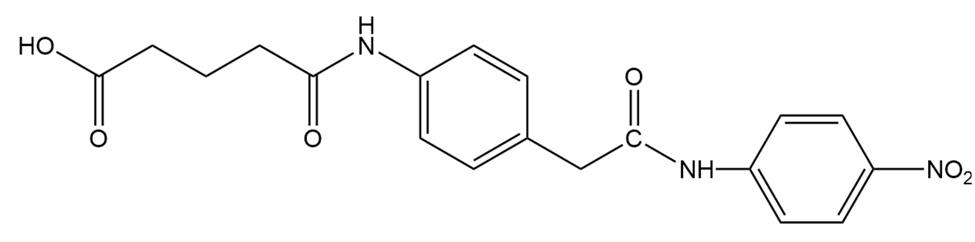
c. Design a transition state analog that catalyzes amide hydrolysis at the amide group indicated.