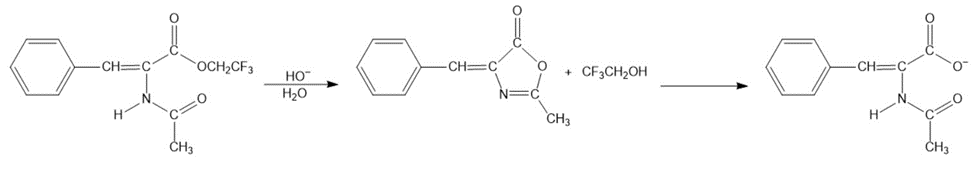
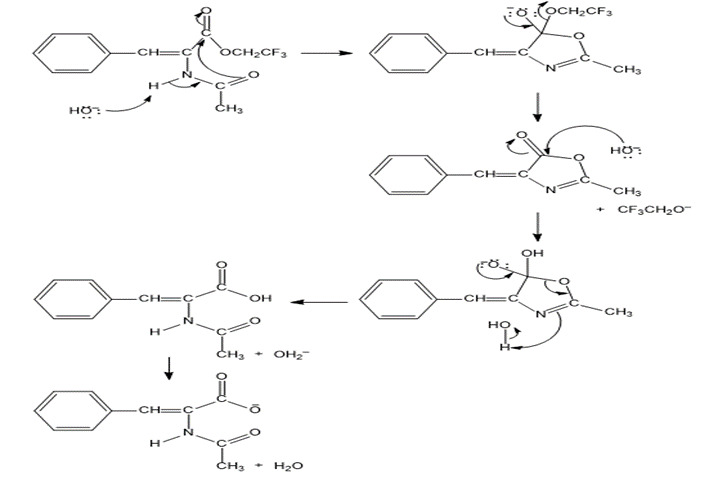
Chapter 15: Q 88p (page 737)
The intermediate shown here is formed during the hydroxide-ion-promoted hydrolysis of ester group. Propose a mechanism for the reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 15: Q 88p (page 737)
The intermediate shown here is formed during the hydroxide-ion-promoted hydrolysis of ester group. Propose a mechanism for the reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
There are three carbon–oxygen bonds in methyl acetate.
a.What are their relative bond lengths?
b.What are the relative infrared (IR) stretching frequencies of these bonds?
When butanoic acid and 18O-labelled methanol react under acidic conditions, what compounds are labelled when the reaction has reached equilibrium?
Write the mechanism for the acid-catalyzed reaction of an amide with an alcohol to form an ester.
Draw the structure for each of the following:
\(\begin{array}{}a.{\rm{ phenyl acetate}}\\b.{\rm{ }}\gamma {\rm{ - caprolactam}}\\c.{\rm{ sodium formate}}\\d.{\rm{ N - benzylethanamide}}\\e.{\rm{ }}\gamma {\rm{ - methylcaproic acid}}\\f.{\rm{ }}\beta {\rm{ - bromobutyramide}}\\g.{\rm{ ethyl 2 - chloropentanoate}}\\h.{\rm{cyclohexanecarbonyl chloride}}\\i.{\rm{ }}\alpha {\rm{ - chlorovaleric acid}}\\\end{array}\)
How could you use 1H NMR spectroscopy to distinguish the following esters?
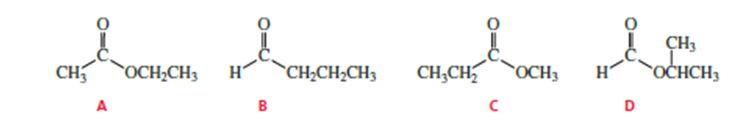
What do you think about this solution?
We value your feedback to improve our textbook solutions.