Chapter 15: Q35P (page 715)
Write the mechanism for the acid-catalyzed reaction of an amide with an alcohol to form an ester.
Learning Materials
Features
Discover
Chapter 15: Q35P (page 715)
Write the mechanism for the acid-catalyzed reaction of an amide with an alcohol to form an ester.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why, in the last step of the mechanism for hydroxide-ion promoted hydrolysis of an amide, is the amide ion protonated?
1,4-Diazabicyclo [2.2.2] octane (abbreviated DABCO) is a tertiary amine that catalyzes transesterification reactions. Explain how it does this.
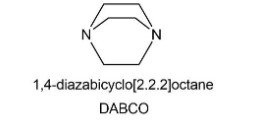
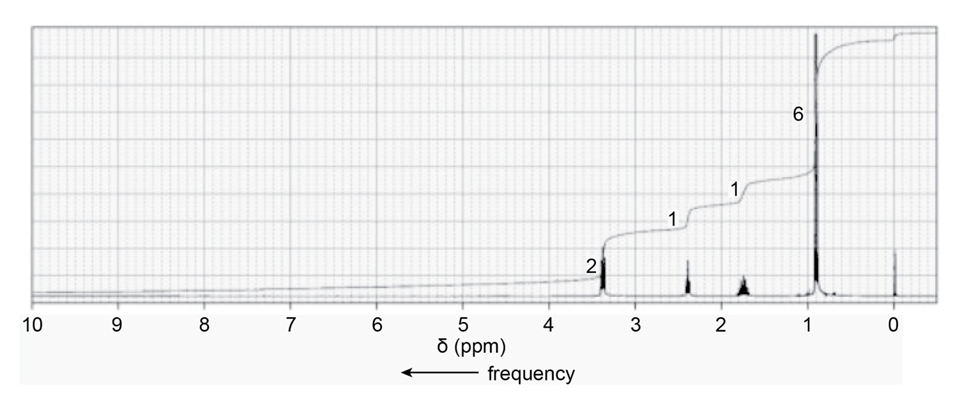
When a compound with molecular formula undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following NMR spectrum. Identify the compound.
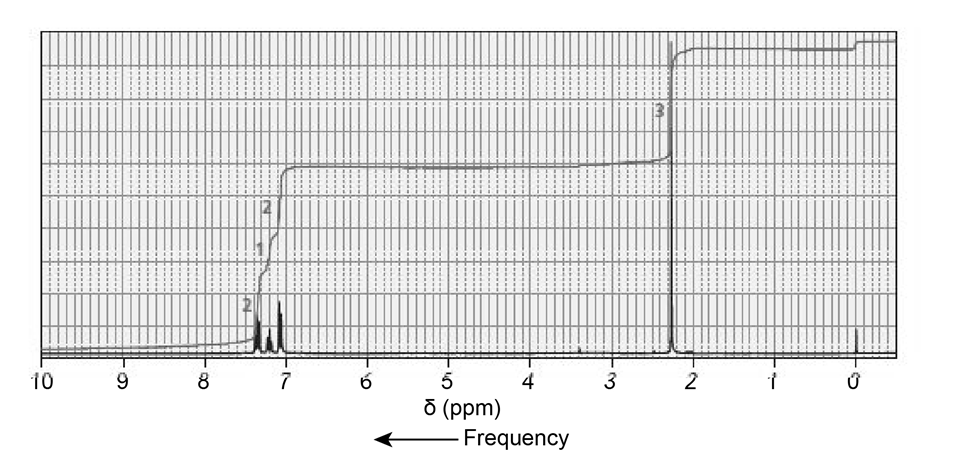
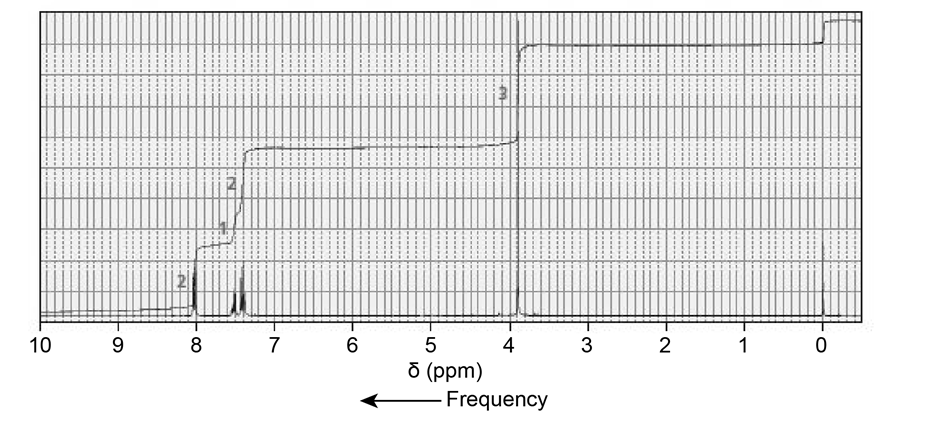
The NMR spectra for two esters with molecular formula are shown next. Which of the esters is hydrolyzed more rapidly in an aqueous solution with a pH of 10?
The aromas of many flowers and fruits are due to esters such as those shown in this problem. What are the common names of these esters? (Also see Problem 57.)
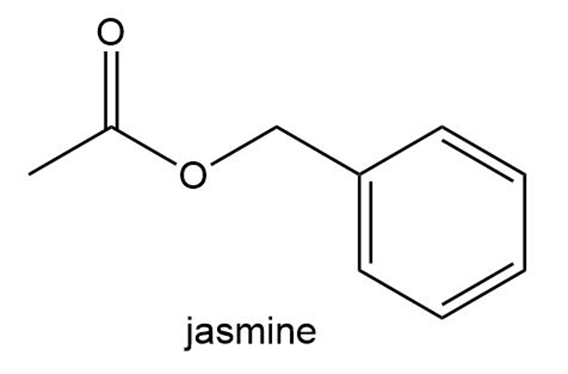
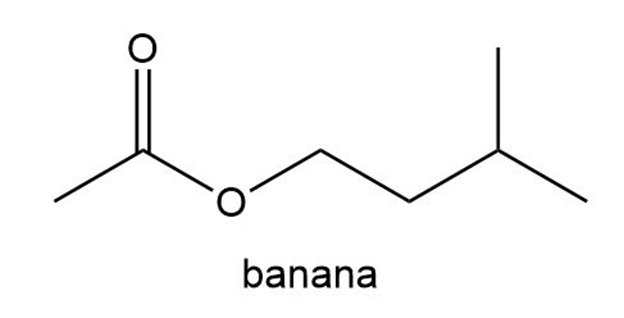
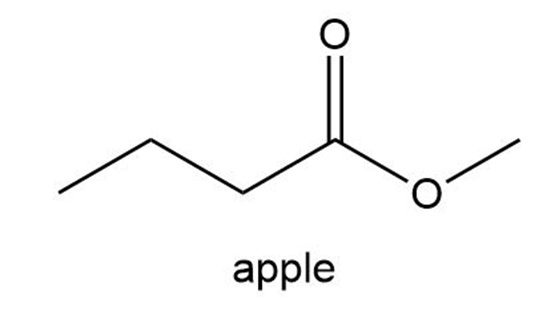
What do you think about this solution?
We value your feedback to improve our textbook solutions.