Chapter 15: Q84P (page 737)
The NMR spectra for two esters with molecular formula are shown next. Which of the esters is hydrolyzed more rapidly in an aqueous solution with a pH of 10?
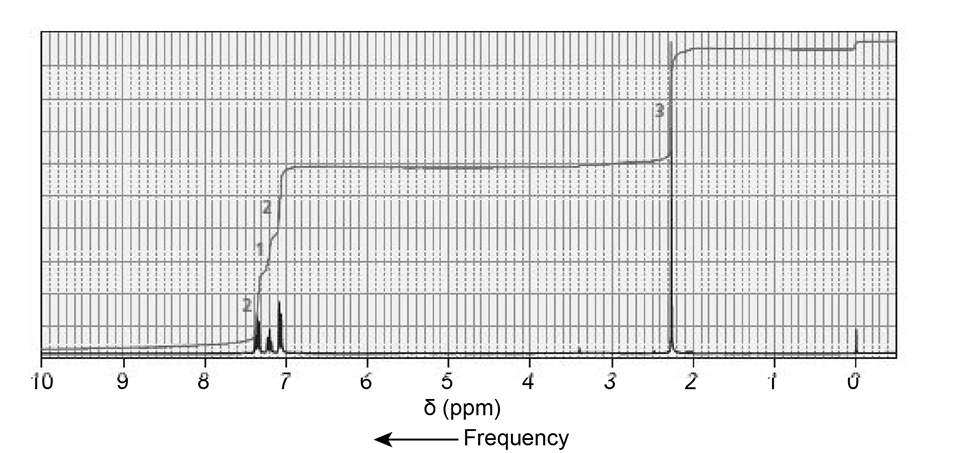
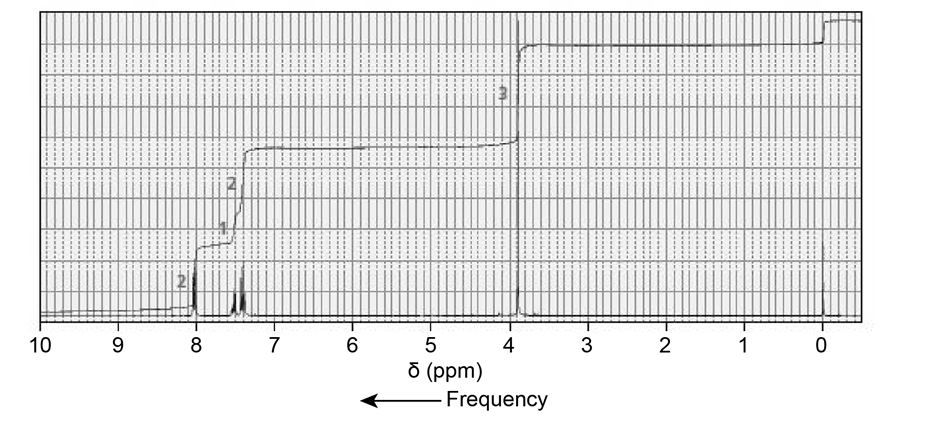
Short Answer
Following ester will hydrolyze more rapidly:
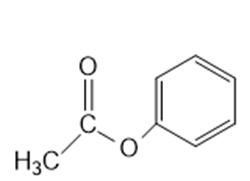
Learning Materials
Features
Discover
Chapter 15: Q84P (page 737)
The NMR spectra for two esters with molecular formula are shown next. Which of the esters is hydrolyzed more rapidly in an aqueous solution with a pH of 10?


Following ester will hydrolyze more rapidly:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw a structure for each of the following:
a. N,N-dimethylhexanamide
b. 3,3-dimethylhexanamide
c. Cyclohexanecarbonyl chloride
d. Propanenitrile
e. propionamide
f. sodium acetate
g. benzoic anhydride
h. b-valerolactone
i. 3-methylbutanenitrile
j. cycloheptanecarboxylic acid
k. benzoyl chloride
Phosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the following reagents?
a. one equivalent of methanol
b. excess methanol
c. excess propylamine
d. excess water
What alkyl bromide would you use in a Gabriel synthesis to prepare each of the following amines?
Pentylamine
Iso hexyl amine
Benzylamine
Cyclohexylamine
Propose a mechanism that accounts for the formation of the product.
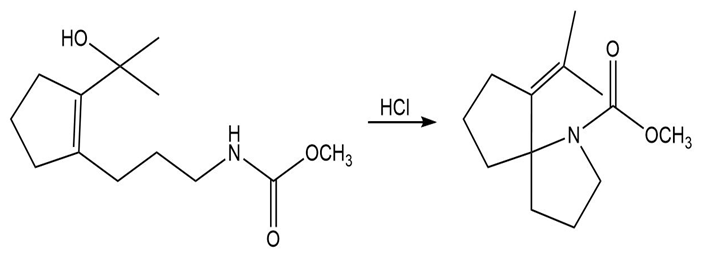
The following compound has been found to be an inhibitor of penicillinase. The enzyme can be reactivated by hydroxylamine . Propose a mechanism to account for the inhibition and for the reactivation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.