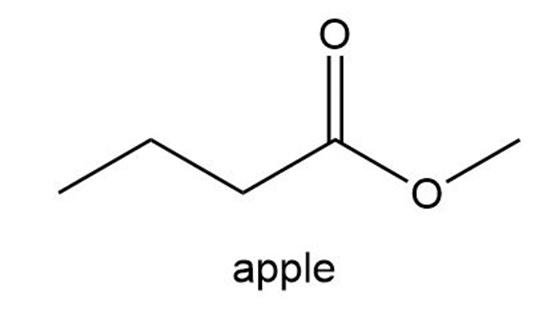
Chapter 15: Q69P (page 735)
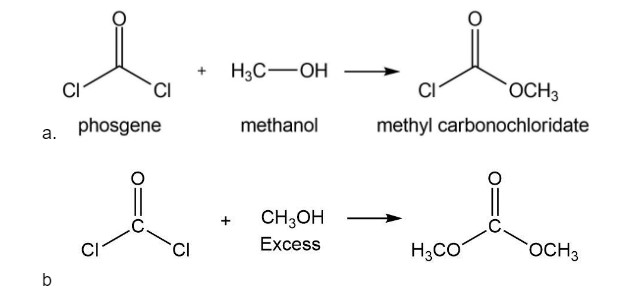
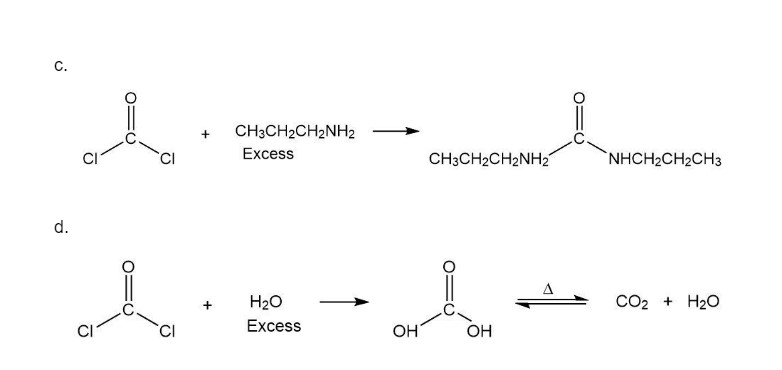
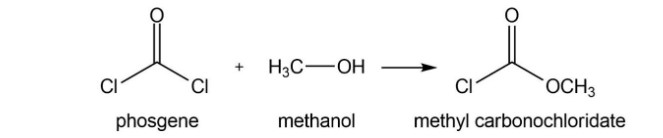
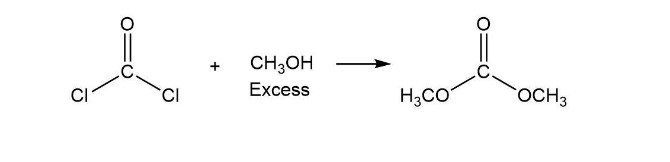
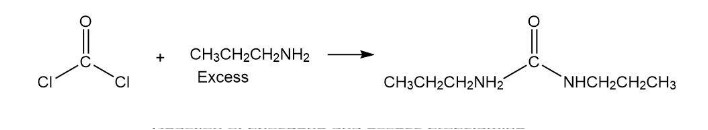
Phosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the following reagents?
a. one equivalent of methanol
b. excess methanol
c. excess propylamine
d. excess water
Short Answer