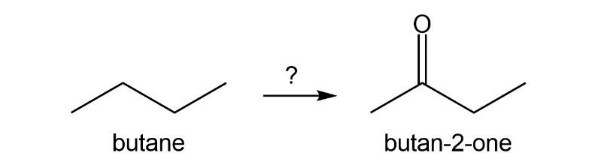
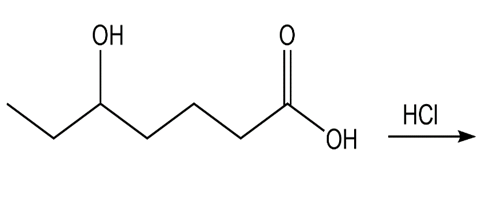
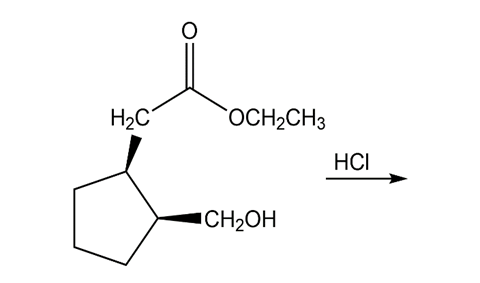
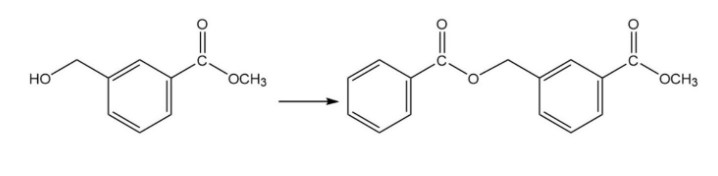
Chapter 15: Q7P (page 696)
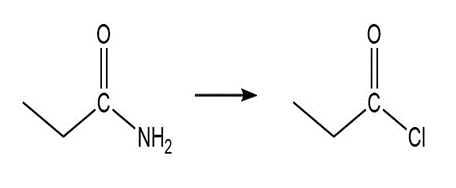
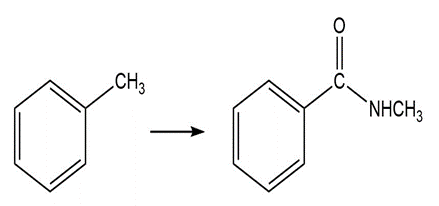
a. What is the product of the reaction of acetyl chloride with? Theof HCl is -7; theofis 15.7.
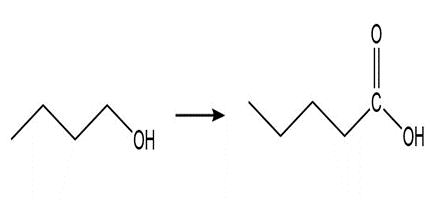
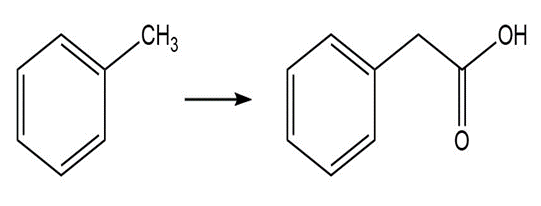
b. What is the product of the reaction of acetamide with? Theofis 36; theofis 15.7.
Short Answer
a.
 Formation of acetic acid
Formation of acetic acid
b.
 No products formed
No products formed
 Formation of acetic acid
Formation of acetic acid No products formed
No products formed