Chapter 15: Q64P (page 733)
Describe how the target molecule (butanone) can be synthesized in a high yield from butane.
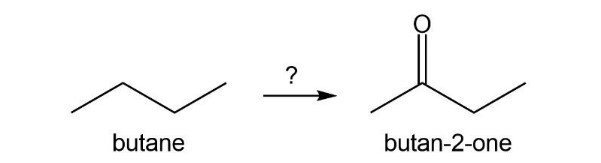
Short Answer
Reaction followed as:
Learning Materials
Features
Discover
Chapter 15: Q64P (page 733)
Describe how the target molecule (butanone) can be synthesized in a high yield from butane.

Reaction followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
Why, in the last step of the mechanism for hydroxide-ion promoted hydrolysis of an amide, is the amide ion protonated?
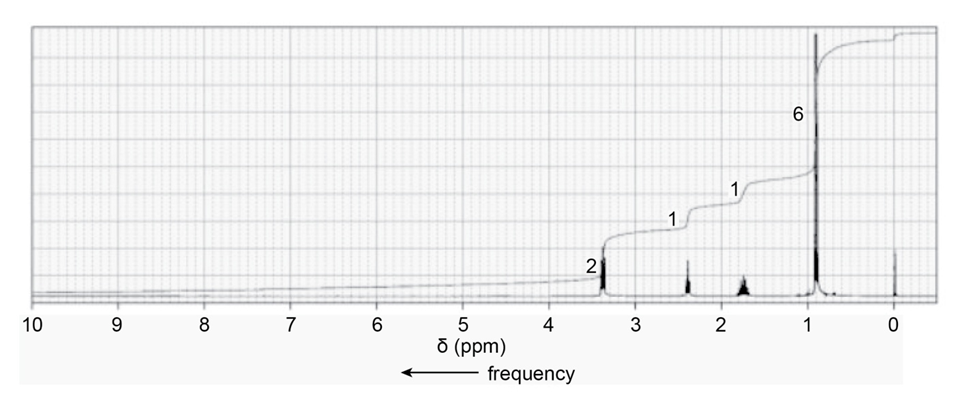
When a compound with molecular formula undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following NMR spectrum. Identify the compound.
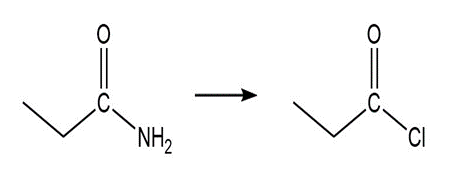
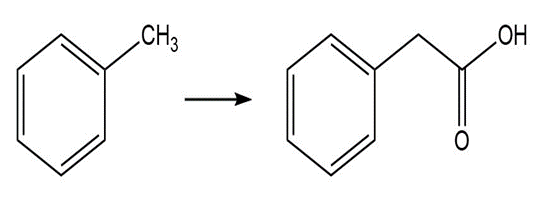
Question: Show how the following compounds could be prepared from the given starting materials. You can use any necessary organic or inorganic reagents.
a.
b.
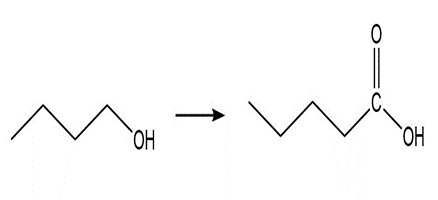
c.
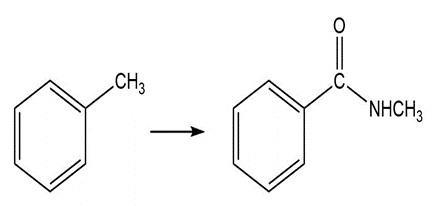
d.
When a student treated butane dioic acid with thionyl chloride, she was surprised to find that the product she obtained was an anhydride rather than an acyl chloride. Propose a mechanism to explain why she obtained an anhydride.
How could you synthesize the following compounds starting with a carboxylic acid?
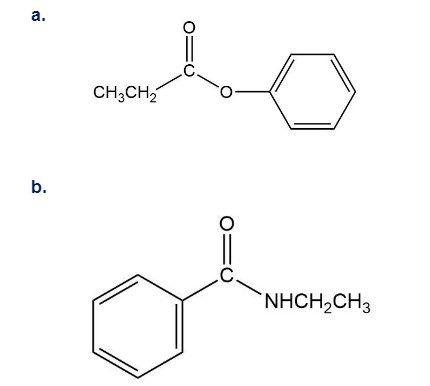
What do you think about this solution?
We value your feedback to improve our textbook solutions.