Chapter 15: Q47P (page 724)
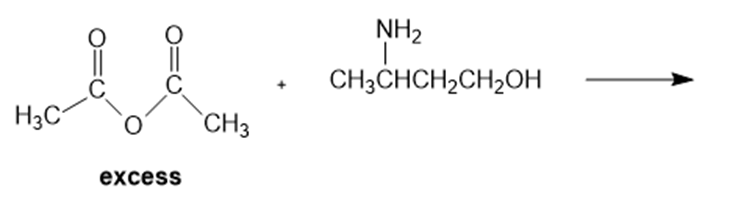
How could you synthesize the following compounds starting with a carboxylic acid?
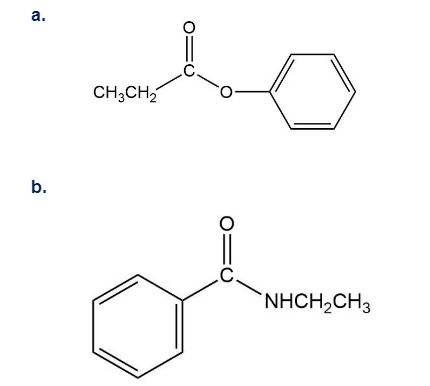
Short Answer
The reaction followed as:
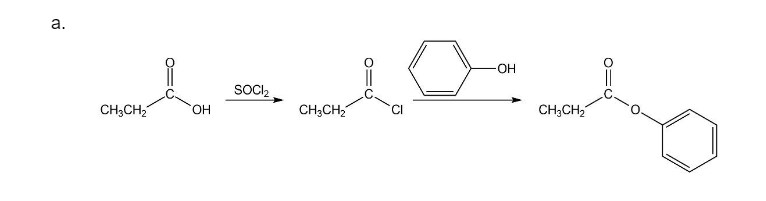
b.
Learning Materials
Features
Discover
Chapter 15: Q47P (page 724)
How could you synthesize the following compounds starting with a carboxylic acid?

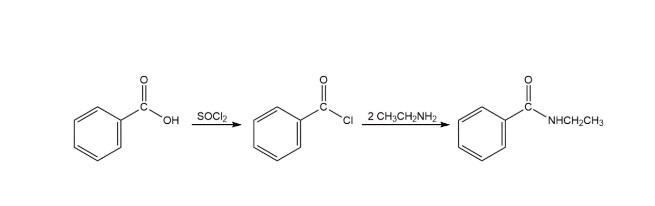
The reaction followed as:

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how the following compounds could be prepared from the given starting materials. You can use any necessary organic or inorganic reagents.
a.
b.
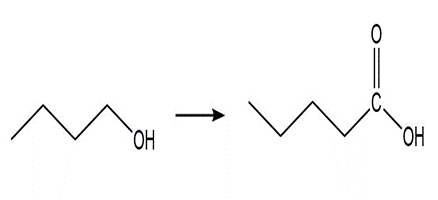
c.
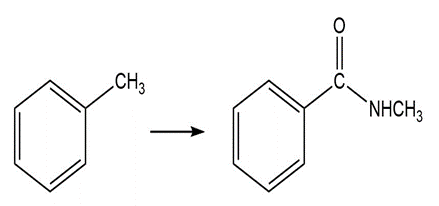
d.
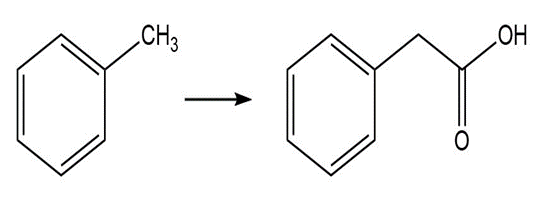
Write the mechanism for the acid-catalyzed reaction of an amide with an alcohol to form an ester.
Information about the mechanism of the reaction undergone by a series of substituted benzenes can be obtained by plotting logarithm of the observed rate constant determined at particular pH against the Hammett substituent constant for the particular substituent. The value for hydrogen is zero. Electron-donating substituents have negative values; the more strongly electron withdrawing the substituent, the more positiveits s value. The slope of a plot of the logarithm of the rate constant versusis called the (rho) value. The value for the hydroxide-ion-promoted hydrolysis of a series of meta- and para-substituted ethyl
benzoates is ; the value for amide formation for the reaction of a series of meta- and para-substituted anilines with benzoyl chloride is .
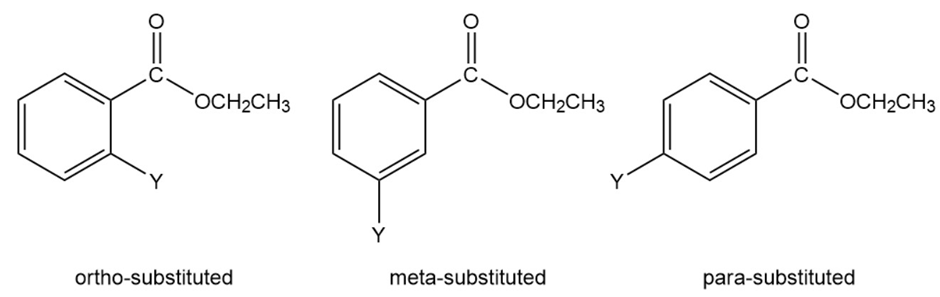
a.Identify the two products obtained from the following reaction:
b. A student carried out the preceding reaction, but stopped it before it was half over, whereupon he isolated the major product. He was surprised to find
that the product he isolated was neither of the products obtained when the reaction was allowed to go to completion. What product did he isolate?
The aromas of many flowers and fruits are due to esters such as those shown in this problem. What are the common names of these esters? (Also see Problem 57.)
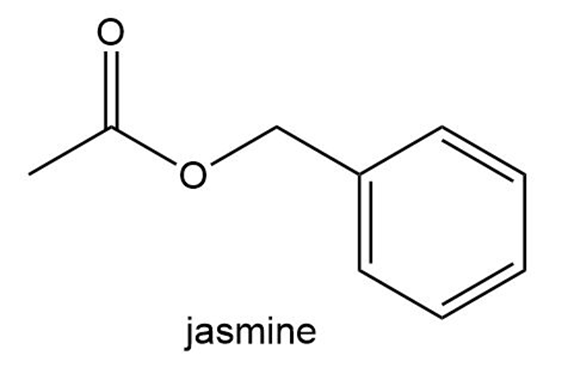
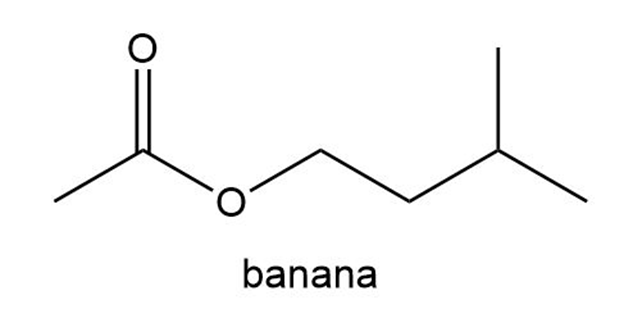
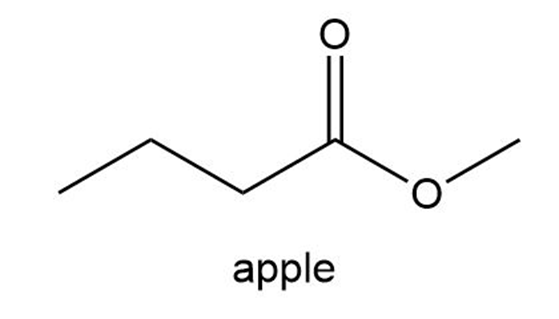
What do you think about this solution?
We value your feedback to improve our textbook solutions.