Chapter 15: Q46P (page 723)
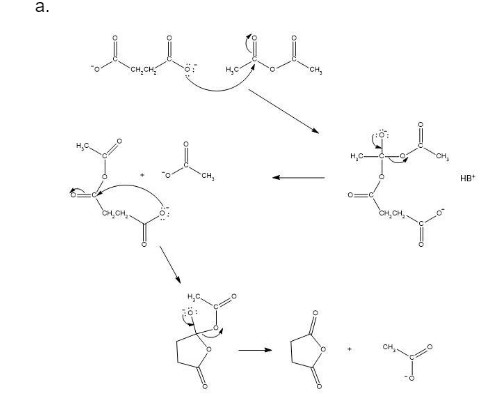
a. Propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride.
b. How does acetic anhydride make it easier to form the anhydride?
Short Answer
Mechanism followed as:
Learning Materials
Features
Discover
Chapter 15: Q46P (page 723)
a. Propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride.
b. How does acetic anhydride make it easier to form the anhydride?
Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
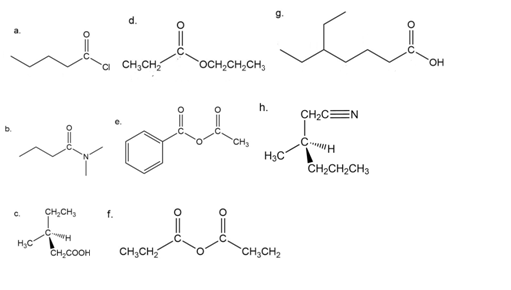
Name the following.
a. Propose a mechanism for the reaction of acetic anhydride with water.
b. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?
The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, does not work with primary alcohols.
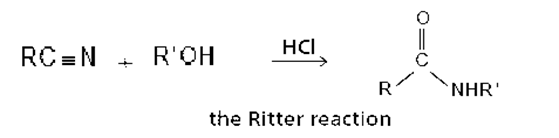
a. Propose a mechanism for the Ritter reaction.
b. Why does the Ritter reaction not work with primary alcohols?
c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?
Draw the structure for each of the following:
\(\begin{array}{}a.{\rm{ phenyl acetate}}\\b.{\rm{ }}\gamma {\rm{ - caprolactam}}\\c.{\rm{ sodium formate}}\\d.{\rm{ N - benzylethanamide}}\\e.{\rm{ }}\gamma {\rm{ - methylcaproic acid}}\\f.{\rm{ }}\beta {\rm{ - bromobutyramide}}\\g.{\rm{ ethyl 2 - chloropentanoate}}\\h.{\rm{cyclohexanecarbonyl chloride}}\\i.{\rm{ }}\alpha {\rm{ - chlorovaleric acid}}\\\end{array}\)
a. What is the product of the reaction of acetyl chloride with? Theof HCl is -7; theofis 15.7.
b. What is the product of the reaction of acetamide with? Theofis 36; theofis 15.7.
What do you think about this solution?
We value your feedback to improve our textbook solutions.