Chapter 15: Q70P (page 735)
What reagent should be used to carry out the following reaction?
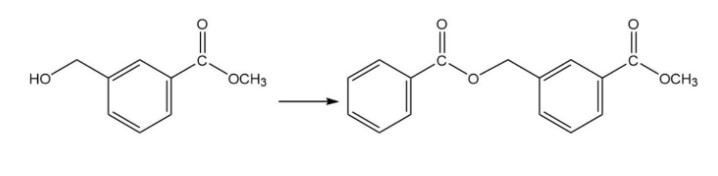
Short Answer
The reagent used is benzoyl chloride
Learning Materials
Features
Discover
Chapter 15: Q70P (page 735)
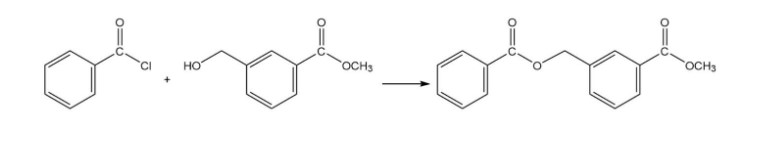
What reagent should be used to carry out the following reaction?

The reagent used is benzoyl chloride

All the tools & learning materials you need for study success - in one app.
Get started for free
Which is a correct statement?
A. The delocalization energy of an ester is about 18 kcal/mol, and the delocalization energy of an amide is about 10 kcal/mol.
B. The delocalization energy of an ester is about 10 kcal/mol, and the delocalization energy of an amide is about 18 kcal/mol.
The NMR spectra for two esters with molecular formula are shown next. Which of the esters is hydrolyzed more rapidly in an aqueous solution with a pH of 10?
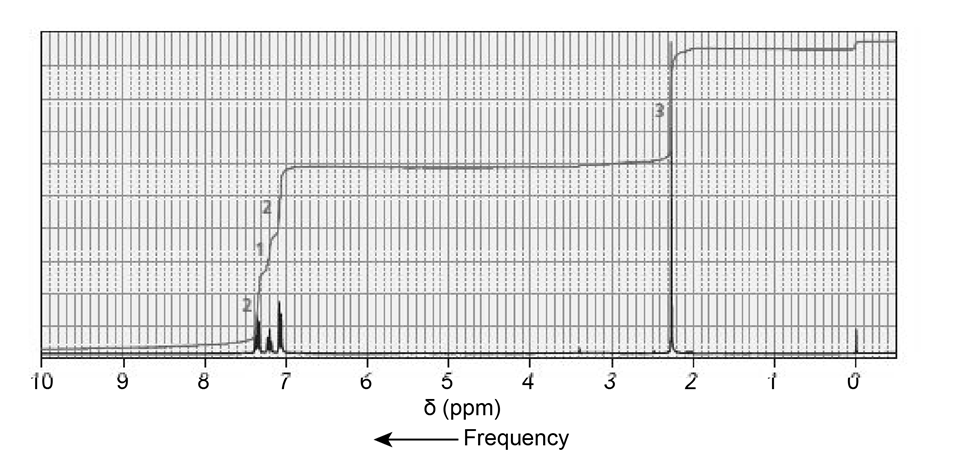
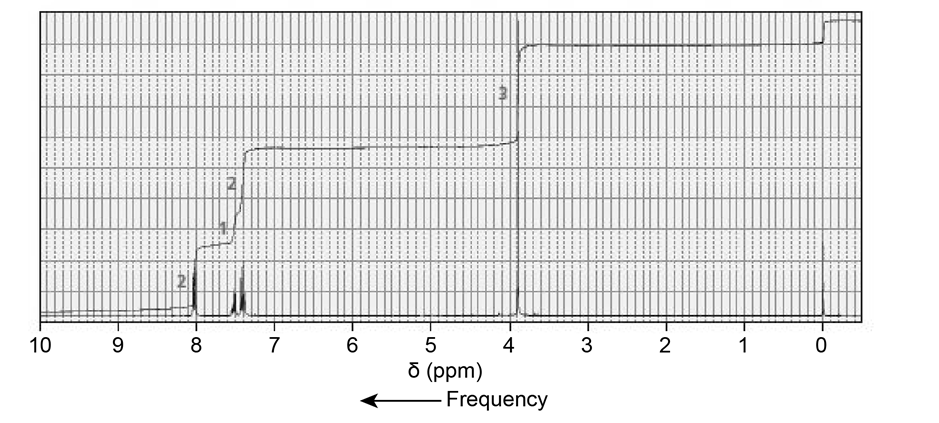
Phosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the following reagents?
a. one equivalent of methanol
b. excess methanol
c. excess propylamine
d. excess water
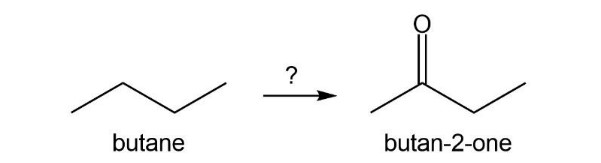
Describe how the target molecule (butanone) can be synthesized in a high yield from butane.
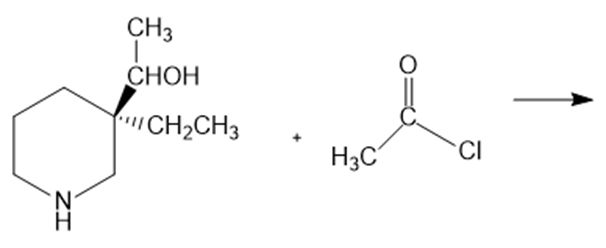
Identify the major and minor products of the following reaction:
What do you think about this solution?
We value your feedback to improve our textbook solutions.