Chapter 15: Q86P (page 737)
Question: Is the acid-catalyzed hydrolysis of acetamide a reversible or an irreversible reaction? Explain.
Short Answer
Answer
Acid-catalyzed hydrolysis of acetamide is an irreversible reaction.
Learning Materials
Features
Discover
Chapter 15: Q86P (page 737)
Question: Is the acid-catalyzed hydrolysis of acetamide a reversible or an irreversible reaction? Explain.
Answer
Acid-catalyzed hydrolysis of acetamide is an irreversible reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
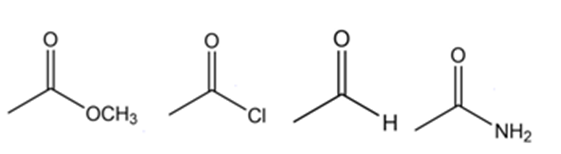
Rank the following compounds in order of decreasing frequency of the carbon-oxygen double bond stretch
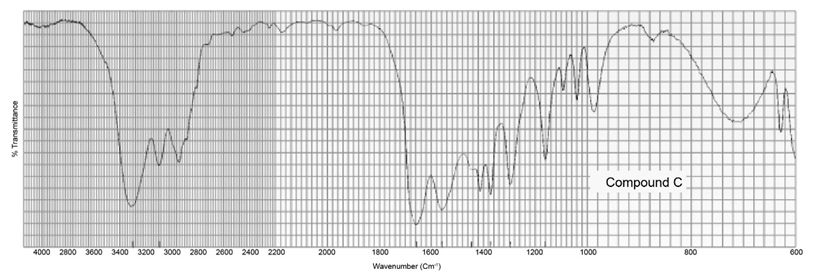
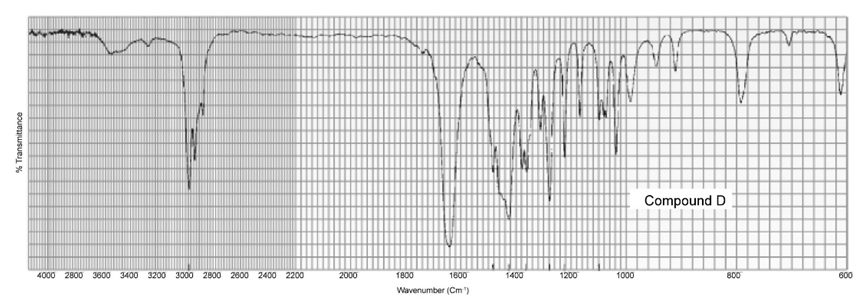
Two products, A and B, are obtained from the reaction of 1-bromobutane with NH3 . Compound A reacts with acetyl chloride to form C, and compound B reacts with acetyl chloride to form D. The IR spectra of C and D are shown. Identify A, B, C, and D.
Question: Draw a structure for each of the following:
a. N,N-dimethylhexanamide
b. 3,3-dimethylhexanamide
c. Cyclohexanecarbonyl chloride
d. Propanenitrile
e. propionamide
f. sodium acetate
g. benzoic anhydride
h. b-valerolactone
i. 3-methylbutanenitrile
j. cycloheptanecarboxylic acid
k. benzoyl chloride
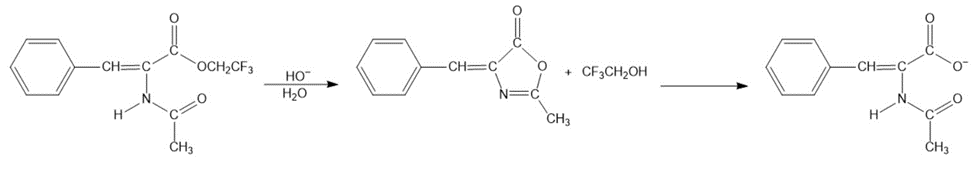
The intermediate shown here is formed during the hydroxide-ion-promoted hydrolysis of ester group. Propose a mechanism for the reaction.
Which is longer, the carbon–oxygen single bond in a carboxylic acid or the carbon–oxygen bond in an alcohol? Why?
What do you think about this solution?
We value your feedback to improve our textbook solutions.