Chapter 16: Q92P (page 799)
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the followingNMR spectrum. Identify the compound.
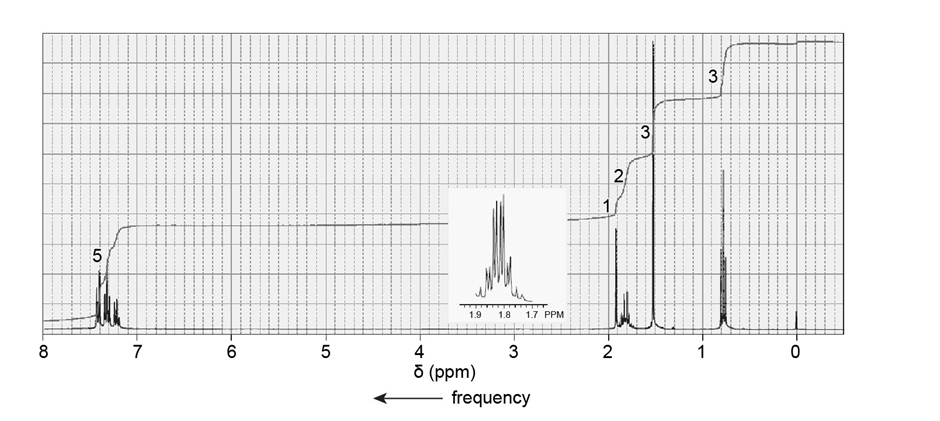
Short Answer
The compound is 1-phenyl-1-propanone.
Learning Materials
Features
Discover
Chapter 16: Q92P (page 799)
A compound reacts with methylmagnesium bromide followed by acidification to form the product with the followingNMR spectrum. Identify the compound.

The compound is 1-phenyl-1-propanone.
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Show how the following compounds can be prepared, using ethyne as one of the starting materials:
b. Explain why ethyne should be alkylated before, rather than after, nucelophilic addition.
Question:What products would be formed from the preceeding reaction if the carboxylic acid group were not protected?
How could you convert N-methylbenzamide to the following compounds?
Why are numbers not used to designate the position of the functional group in propanone and butanedione?
Propose a mechanism for each of the following reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.