Chapter 16: Q44P (page 773)
Question:What products would be formed from the preceeding reaction if the carboxylic acid group were not protected?
Short Answer
Answer
Product obtained in the reaction is 2-oxohexanedioic acid.
Learning Materials
Features
Discover
Chapter 16: Q44P (page 773)
Question:What products would be formed from the preceeding reaction if the carboxylic acid group were not protected?
Answer
Product obtained in the reaction is 2-oxohexanedioic acid.
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how the following compounds can be synthesized from cyclohexanol.
a.
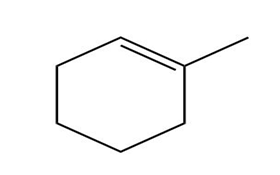
b.

c.

Imines can exist as stereoisomers. The isomers are named using the E, Z system of nomenclature (Section 4.2). The lone pair has the lowest priority.
Draw the structure of each of the following compounds:
a. the (E)-hydrazone of benzaldehyde
b. the (Z)-oxime of propiophenone
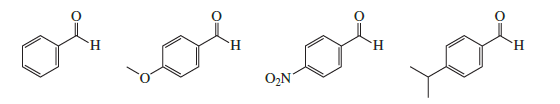
Rank the following compounds from most reactive to least reactive toward nucleophilic addition:
Which of the following are
a. hemiacetals?
b. acetals?
c. hydrates?

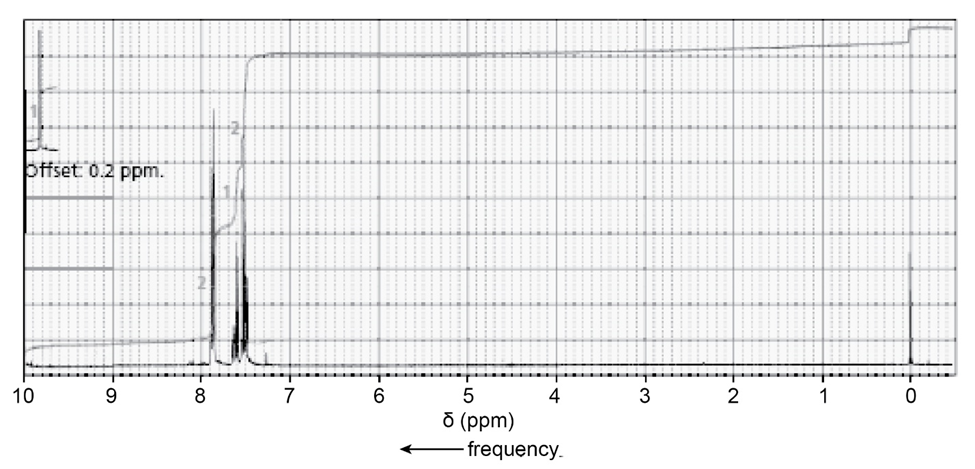
Question: The only organic compound obtained when compound Z undergoes the following sequence of reactions gives the 1H NMR spectrum shown.
Identify compound Z.
What do you think about this solution?
We value your feedback to improve our textbook solutions.