Chapter 16: Q71P (page 795)
Rank the following compounds from most reactive to least reactive toward nucleophilic addition:
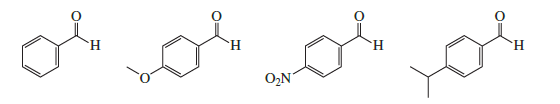
Short Answer
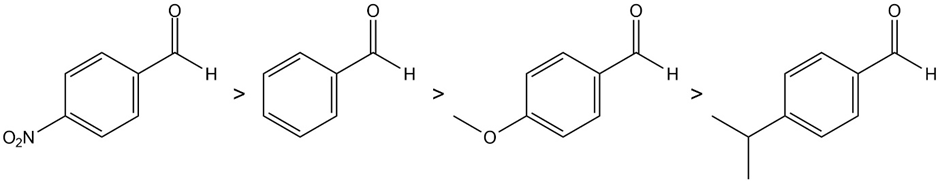
Most reactive least reactive
Order of reaction
Learning Materials
Features
Discover
Chapter 16: Q71P (page 795)
Rank the following compounds from most reactive to least reactive toward nucleophilic addition:


Most reactive least reactive
Order of reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show two ways to convert an alkyl halide into a carboxylic acid that has one more carbon than the alkyl halide.
Draw the products of the following reactions. Indicate whether each reaction is an oxidation or a reduction.
a.
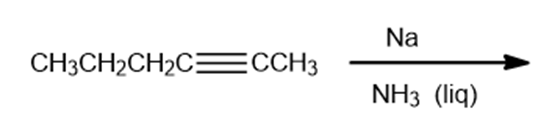
b.
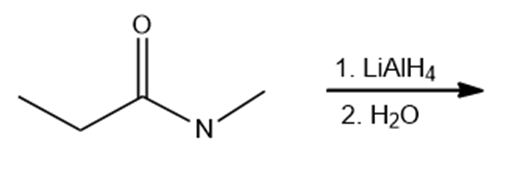
c.
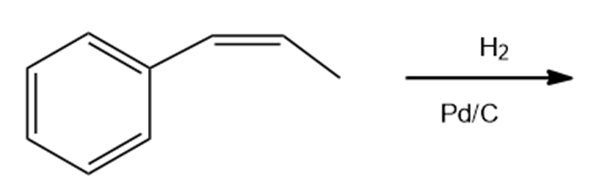
d.
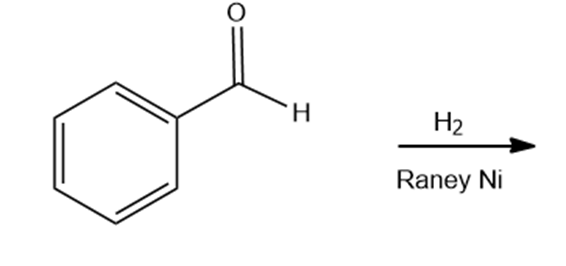
e.
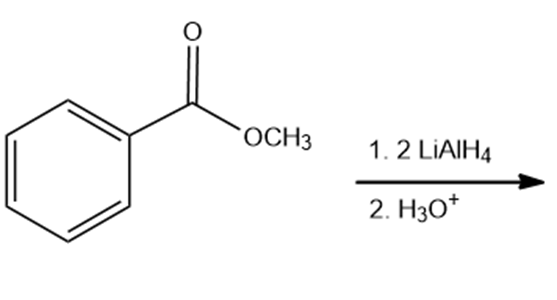
f.
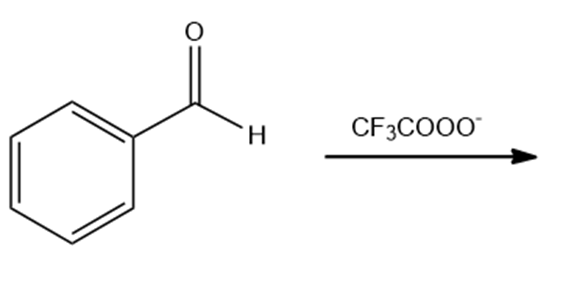
a. Would you expect hemiacetals to be stable in basic solutions? Explain your answer.
b. Acetal formation must be catalyzed by an acid. Explain why it cannot be catalyzed by
c. Can the rate of hydrate formation be increased by hydroxide ion as well as by acid? Explain.
What are the products of the following reactions?
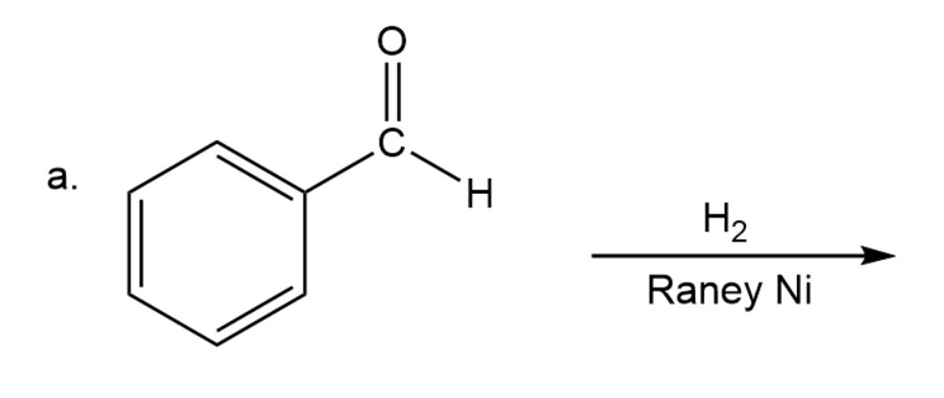
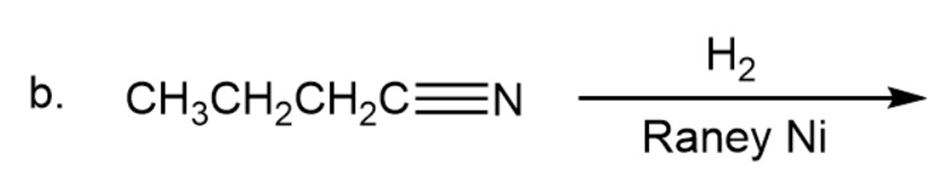
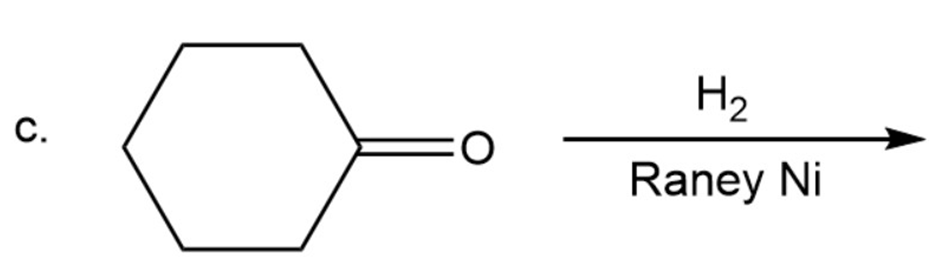
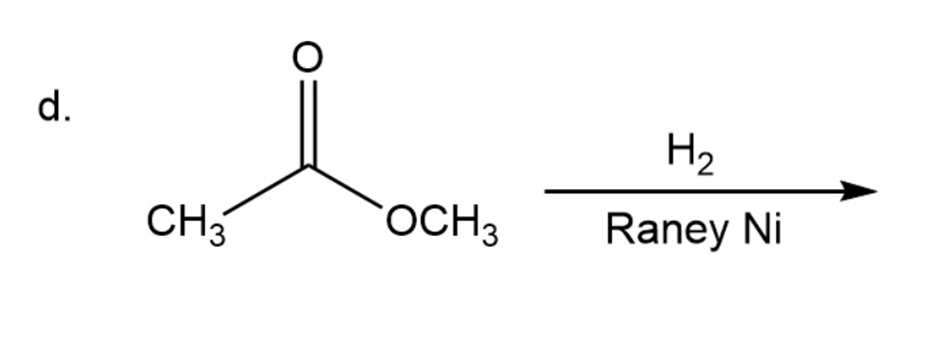
Draw the structures of two esters that will be reduced to propanol and butanol by (followed by addition of aqueous acid).
What do you think about this solution?
We value your feedback to improve our textbook solutions.