Chapter 16: Q19P (page 752)
Question: Show two ways to convert an alkyl halide into a carboxylic acid that has one more carbon than the alkyl halide.
Short Answer
Answer
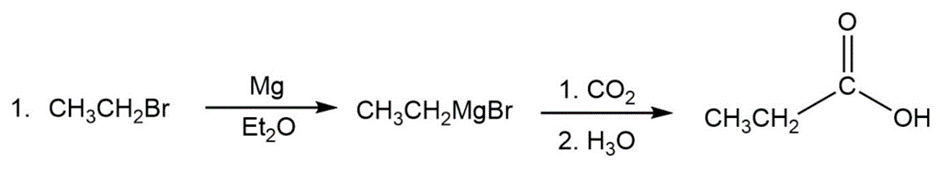
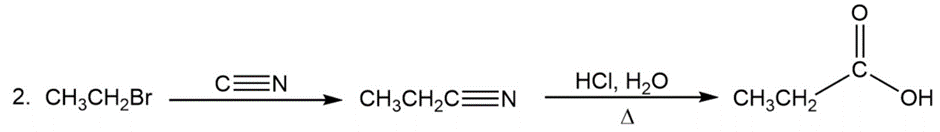
Learning Materials
Features
Discover
Chapter 16: Q19P (page 752)
Question: Show two ways to convert an alkyl halide into a carboxylic acid that has one more carbon than the alkyl halide.
Answer
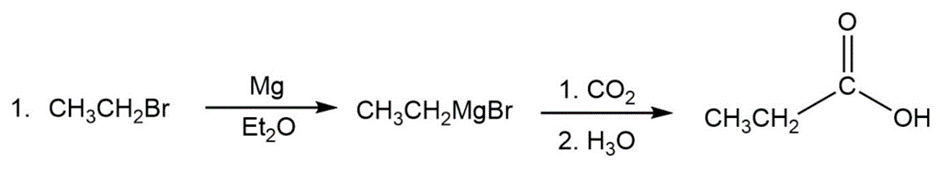
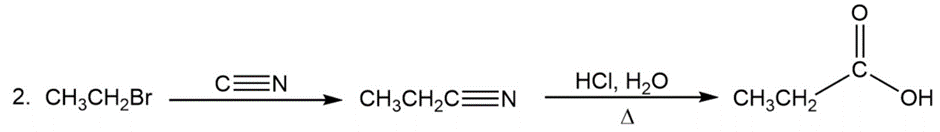
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: a. Show the reagents required to form the primary alcohol in each of the following reactions.b. Which of the reactions cannot be used for the synthesis of isobutyl alcohol?

A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate formed in this reaction, and show how it can be converted to a ketone.
What are the products of the following reactions?
a.
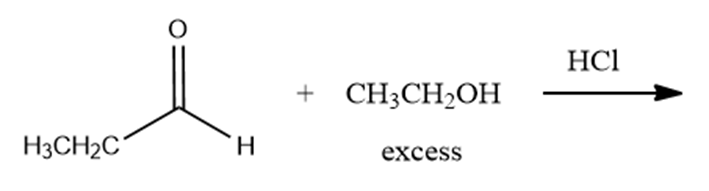
b.
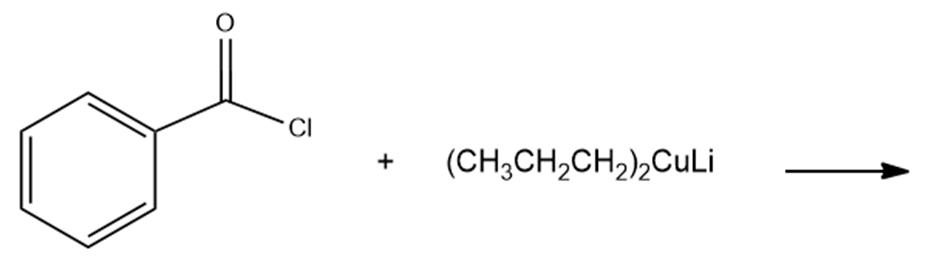
c.
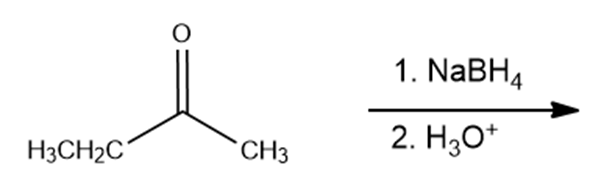
d.
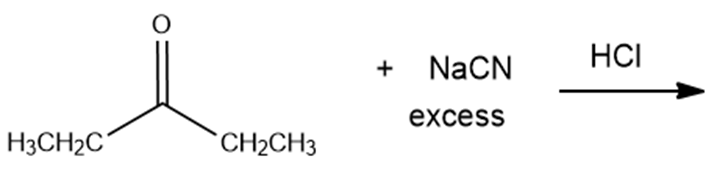
e.
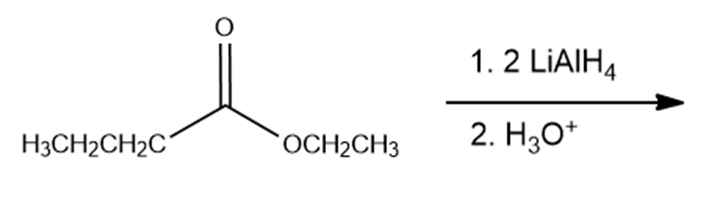
f.
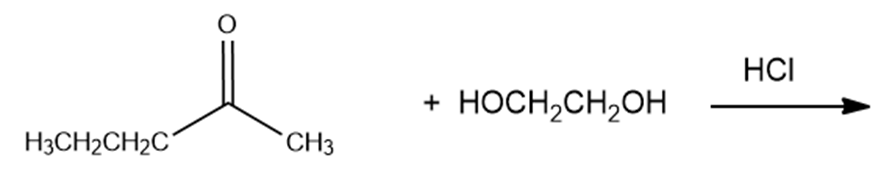
g.
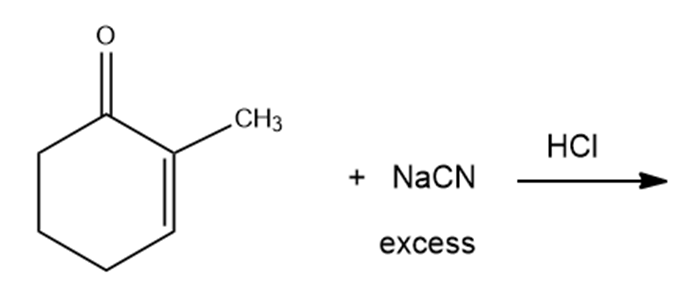
h.
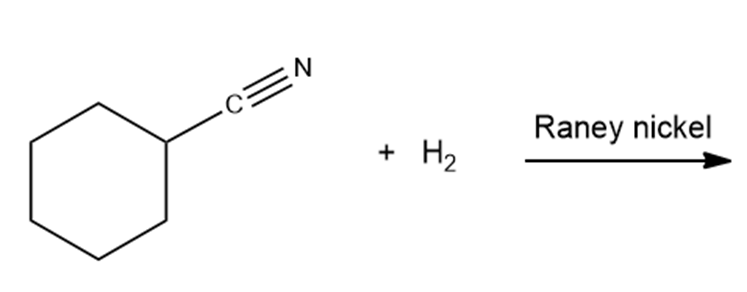
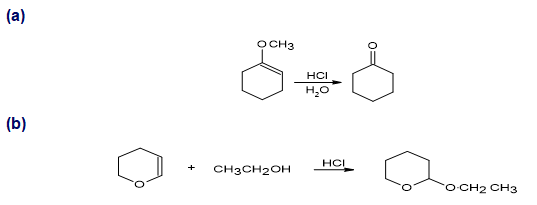
Propose a mechanism for each of the following reactions:
What amides would you react with LiAlH4 to form the following amines?
a. benzylmethylamine c. diethylamine
b. ethylamine d. triethylamine
What do you think about this solution?
We value your feedback to improve our textbook solutions.