Chapter 16: Q68P (page 795)
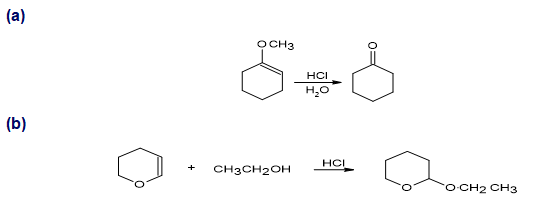
Propose a mechanism for each of the following reactions:
Short Answer
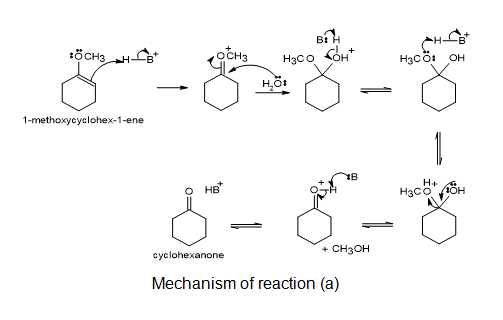
a.
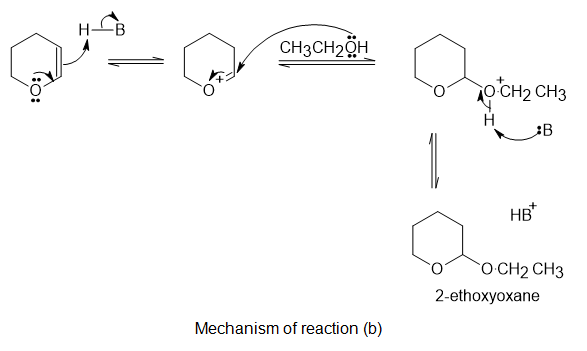
b.
Learning Materials
Features
Discover
Chapter 16: Q68P (page 795)
Propose a mechanism for each of the following reactions:

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis.
a.Propose a mechanism for the reaction.
b.What thiol will be formed if the alkyl halide employed is pentyl bromide?
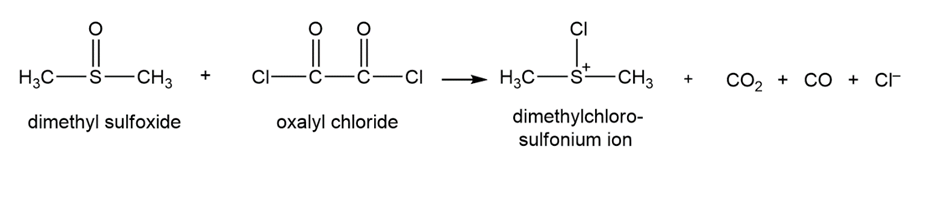
Propose a mechanism to explain how dimethyl sulfoxide and oxalyl chloride react to form the dimethylchlorosulfonium ion used as the oxidizing agent in the Swern oxidation.
What is the product of each of the following reactions?
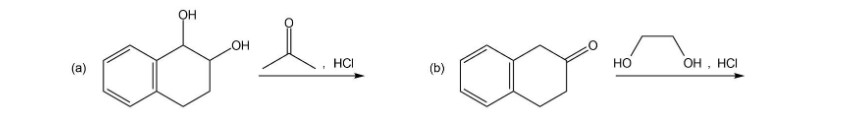
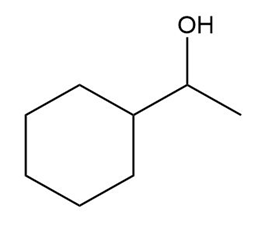
Show how the following compounds can be synthesized from cyclohexanol.
a.
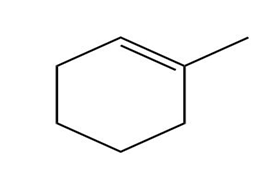
b.
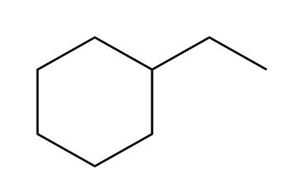
c.
The pKa of protonated acetone is about -7.5, and the pKa of protonated hydroxylamine is 6.0.
a. In a reaction with hydroxylamine at pH 4.5 (Figure 16.2), what fraction of acetone is present in its acidic, protonated form? (Hint: See Section 2.10.)
b. In a reaction with hydroxylamine at pH 1.5, what fraction of acetone is present in its acidic, protonated form?
c. In a reaction with acetone at pH 1.5 (Figure 16.2), what fraction of hydroxylamine is present in its reactive basic form?
What do you think about this solution?
We value your feedback to improve our textbook solutions.