Chapter 16: Q22P (page 757)
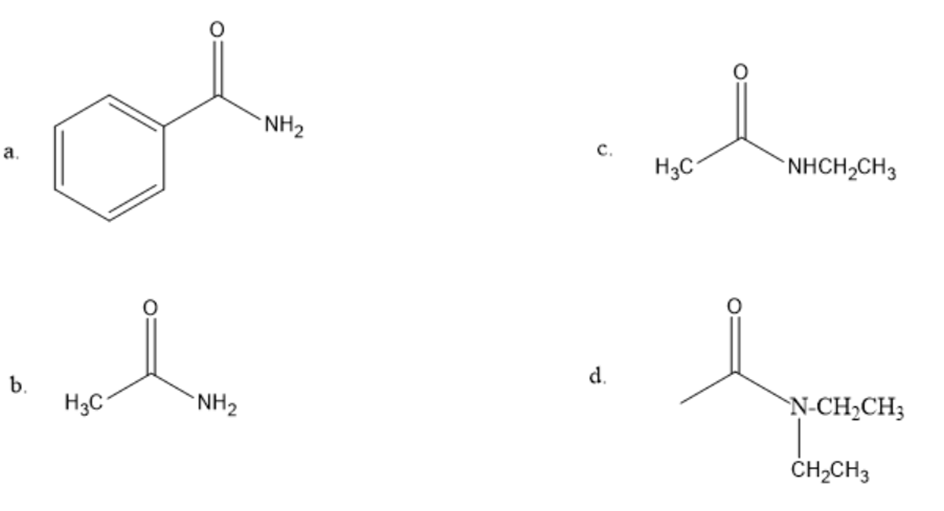
What amides would you react with LiAlH4 to form the following amines?
a. benzylmethylamine c. diethylamine
b. ethylamine d. triethylamine
Short Answer
Learning Materials
Features
Discover
Chapter 16: Q22P (page 757)
What amides would you react with LiAlH4 to form the following amines?
a. benzylmethylamine c. diethylamine
b. ethylamine d. triethylamine

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for each of the following reactions:

Question:Explain why aldehydes and ketones react with a weak acid such as hydrogen cyanide but do not react with strong acids such as HCl or H2SO4 (other than being protonated by them).
Question: Draw the structure for each of the following:
a.
b.
c.
d.
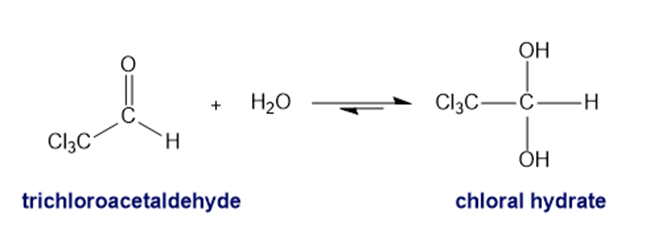
When trichloroacetaldehyde is dissolved in water, almost all of it is converted to the hydrate. Chloral hydrate, the product of the reaction, is a sedative that can be lethal. A cocktail laced with it is known—in detective novels, at least—as a "Mickey Finn." Explain why an aqueous solution of trichloroacetaldehyde is almost all hydrate.
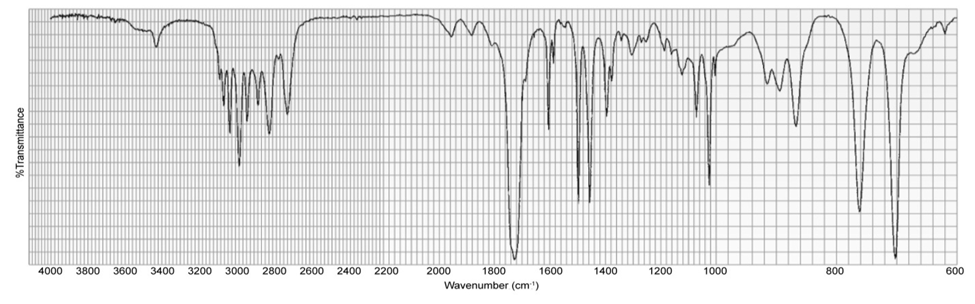
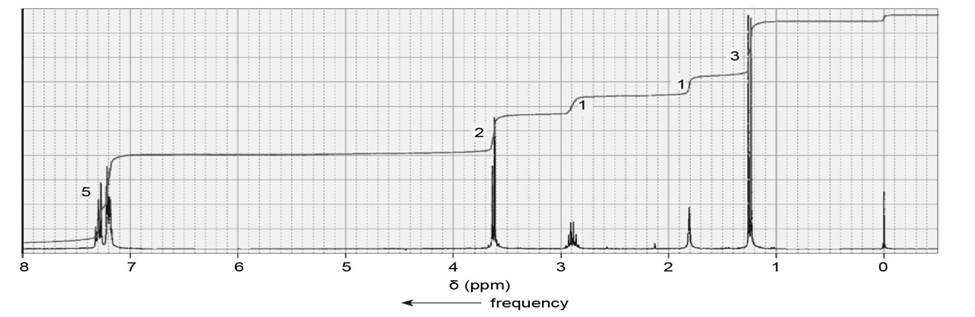
A compound gives the following IR spectrum. Upon reaction with sodium borohydride followed by acidification, it forms the product with the NMR spectrum shown below. Identify the starting material and the product.
What do you think about this solution?
We value your feedback to improve our textbook solutions.